FDA’s CDRH Circulatory System Devices Panel of the Medical Devices Advisory Committee will meet later this year to examine the safety of endovascular stent grafts made by Endologix and other companies. Day one of the meeting will examine the benefit-risk profile of the Endologix AFX endovascular graft system regarding a potentially … [Read more...] about FDA committee to examine safety of endovascular stent grafts
FDA approves Medtronic’s Harmony transcatheter pulmonary valve system
FDA has approved a Medtronic device described as a first-in-the-world, non-surgical heart valve to treat severe pulmonary valve regurgitation — a condition often arising from congenital heart disease. The Harmony transcatheter pulmonary valve system could delay the time before someone with congenital heart disease might need additional … [Read more...] about FDA approves Medtronic’s Harmony transcatheter pulmonary valve system
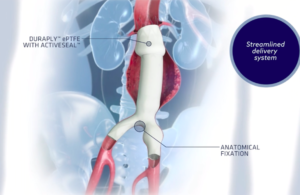
Cook Medical wins FDA breakthrough device designation for next-gen endovascular graft
Cook Medical announced that FDA has granted breakthrough device designation for its Zenith Fenestrated+ endovascular graft (ZFEN+), the next-gen version of its Zenith Fenestrated AAA endovascular graft. The designation — a first for Bloomington, Ind.–based Cook Medical — will enable priority review and better communication with FDA during the … [Read more...] about Cook Medical wins FDA breakthrough device designation for next-gen endovascular graft
Alleviant Medical wins FDA breakthrough device designation for transcath heart failure treatment
Alleviant Medical (Austin, Texas) has received FDA breakthrough device designation for its no-implant, interatrial shunt technology for treating certain types of chronic heart failure. Founded in 2017, Alleviant's minimally invasive transcatheter tech decompresses the left atrium without a permanent cardiac implant or open-heart surgery. … [Read more...] about Alleviant Medical wins FDA breakthrough device designation for transcath heart failure treatment
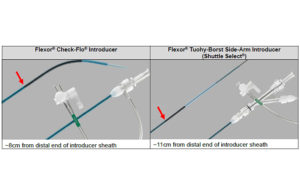
Cook Medical has a serious catheter recall
FDA has designated Cook Medical's recall of its Flexor Check-Flo introducers and Flexor tuohy-borst side-arm introducers as Class I, its most serious level. Cook Medical initiated the recall on Nov. 24. It involves 37,326 of the devices, which are used to deliver medical devices to blood vessels (though not vessels of the heart and brain). The … [Read more...] about Cook Medical has a serious catheter recall
Medtronic has a Class I recall involving balloon catheters to treat heart defects
FDA today designated a Medtronic recall of Rashkind balloon septostomy catheters as Class I, its most serious level. The balloon catheters create an atrial septal defect or to enlarge an existing atrial septal defect in order to treat congenital heart defects. Medtronic (NYSE:MDT) initiated a recall of the Rashkind balloon septostomy … [Read more...] about Medtronic has a Class I recall involving balloon catheters to treat heart defects
J&J wins FDA approval for ablation catheter
Johnson & Johnson (NYSE:JNJ) recently announced that its Biosense Webster business has secured FDA approval for its Thermocool SmartTouch SF ablation catheter. The Thermocool SmartTouch received approval for the treatment of persistent atrial fibrillation, J&J announced yesterday. The approval came after results from the 381-patient, … [Read more...] about J&J wins FDA approval for ablation catheter
These startups are advancing catheter-related medtech
Catheter-related medtech remains an exciting and innovative field — if our sister site Medical Design & Outsourcing's latest list of startups to know is any indication. For example, Neurescue (Copenhagen) touts its aortic occlusion catheter as the first of its type with computer-aided, intelligent safety feedback — and pressure-limited … [Read more...] about These startups are advancing catheter-related medtech
Okami Medical raises $7.2m to support vascular occlusion system
Months after the first use of its LOBO vascular occlusion system on patients, Okami Medical has raised more than $7.2 million. The company's low-profile embolic device features a microcatheter to provide smooth peripheral vasculature navigation. When deployed, the occluder self-expands, conforming to a range of targets such as curved … [Read more...] about Okami Medical raises $7.2m to support vascular occlusion system
Freudenberg Medical completes extractables testing for its PharmaFocus Premium tubing
Freudenberg Medical (Carpinteria, Calif.) today touted extractables testing performed on its PharmaFocus Premium silicone tubing used in biopharma fluid processing and single-use applications. Eurofins Lancaster Laboratories conducted the independent extractables testing for Freudenberg Medical, drawing on BPOG and USP 665 protocols. “We … [Read more...] about Freudenberg Medical completes extractables testing for its PharmaFocus Premium tubing