Micro Medical Solutions today said it has received FDA breakthrough device designation for its MicroStent vascular stent. The stent is designed to maintain vessel patency, enhance wound treatment and improve quality of life and blood flow to reduce amputations and mortality for patients with critical limb-threatening ischemia (CLTI) from … [Read more...] about Micro Medical Solutions wins FDA breakthrough device designation for vascular stent
Perfuze wins CE mark approval for clot aspiration catheter, treats first patients
Perfuze today said it received CE mark approval for its clot aspiration catheter Millipede 088. The Ireland-based company also announced that the first five acute ischemic stroke patients have been treated with the device. “Our initial experience with the device has been very positive, I believe the large bore and distal flow control … [Read more...] about Perfuze wins CE mark approval for clot aspiration catheter, treats first patients
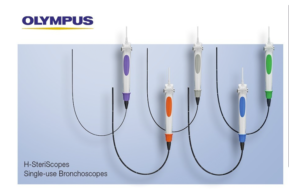
Olympus launches five single-use bronchoscopes
Olympus today said it has launched its product line of five H-SteriScope single-use bronchoscopes. The single-use endoscopes are for diagnostic and therapeutic procedures. They resulted from a collaboration between Olympus subsidiary Veran Medical Technologies and Hunan Vathin Medical Instrument. "We are very excited to launch our first line … [Read more...] about Olympus launches five single-use bronchoscopes
LeMaitre profits up 86% in Q1 results
LeMaitre Vascular (NSDQ:LMAT) posted first-quarter results yesterday that beat the consensus on Wall Street. The company reported profits of $5.9 million, or 28¢ per share, on sales of $35.8 million, for the three months ended March 31, for a bottom-line gain of 86.8% sales growth of 17.45% compared with Q1 2020. Get the full story on our … [Read more...] about LeMaitre profits up 86% in Q1 results
4 catheter-based devices to watch for in 2021
Medical device companies are working to gain regulatory approvals and launch new catheter-based devices as the world slowly returns to normal after reeling from the COVID-19 pandemic. As of April 19, the FDA had approved 11 medical devices this year — two of them catheter-based. In comparison, the regulatory agency approved 19 devices by April … [Read more...] about 4 catheter-based devices to watch for in 2021
This device stops aorta blood flow to save lives
Stopping internal bleeding from torso trauma has its technological challenges. Asha Parekh, co-founder and CEO of Front Line Medical Technologies, has developed a small, quick and easy device that could solve the problem. Front Line Medical Technologies (London, Ontario) is an early-stage medical device company that is developing a way to stop … [Read more...] about This device stops aorta blood flow to save lives
Ambu nabs Health Canada clearance for single-use cystoscope
Ambu today said it received Health Canada clearance for its aScope 4 Cysto single-use cystoscope. The cystoscope is a flexible, single-use endoscope that is designed to eliminate significant capital, repair and cleaning costs with a patient-ready instrument for every procedure, according to Ambu. The device is used for diagnosing, managing and … [Read more...] about Ambu nabs Health Canada clearance for single-use cystoscope
Red One Medical, Quick Tube Medical partner to deliver chest tubes to U.S. military
Red One Medical and Quick Tube Medical this week announced a partnership to provide chest tube devices to U.S. military members. Quick Tube Medical's chest tube system has a rapid deployment system for an average insertion to completion time of 30 to 45 seconds compared to eight minutes for some traditional chest tubes. "This cutting-edge … [Read more...] about Red One Medical, Quick Tube Medical partner to deliver chest tubes to U.S. military
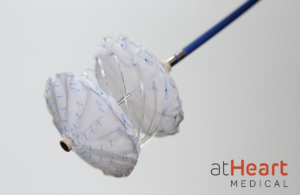
atHeart Medical launches atrial septal defect occluder trial
AtHeart Medical is initiating a U.S. investigational device exemption trial for its atrial septal defects (ASD) occluder. The reSept ASD Occluder is an occluder with a metal-free, bioresorbable frame. The implant is designed to overcome the limitations of occluders by reducing the risk of complications associated with the long-term presence of … [Read more...] about atHeart Medical launches atrial septal defect occluder trial
Seigla Medical gains CE mark for guide catheter extension
Startup Seigla Medical recently announced that it won CE mark approval for its LiquID guide catheter extension for use in coronary and peripheral vasculature interventions. Guide catheter extensions are used by cardiologists to assist in the delivery of stents and other interventional devices, according to the Minneapolis-based … [Read more...] about Seigla Medical gains CE mark for guide catheter extension