BD today announced it started enrollment in the first-in-human trial of a peripheral sirolimus drug-coated balloon (DCB). The Prevision trial is a prospective, multi-center, single-arm, non-randomized study that will evaluate the safety of BD's DCB in the treatment of peripheral arterial disease (PAD) in the femoropopliteal arteries. The company … [Read more...] about BD initiates first-in-human trial of sirolimus drug-coated balloon
The 24 best medical device innovations of 2022
The Galien Foundation recently announced its nominees of medical device innovations for its 2022 Prix Galien USA awards. There are 24 medical technologies nominated for the annual award this year, up from 18 nominees in 2021. The Galien Foundation’s annual Prix Galien awards highlight devices, biotechnology and pharmaceutical products … [Read more...] about The 24 best medical device innovations of 2022
Boston Scientific acquires Obsidio and its gel embolic material
Boston Scientific (NYSE:BSX) today announced the acquisition of Obsidio and its blood vessel embolization material technology. The financial details of the deal were not disclosed. Obsidio develops gel embolic material (GEM) technology that is used for the embolization of blood vessels in the peripheral vasculature. It is a semi-solid, gel-link … [Read more...] about Boston Scientific acquires Obsidio and its gel embolic material
Preceptis Medical wins FDA clearance for expanded indications of Hummingbird tympanostomy tube system
Preceptis Medical today announced its Hummingbird tympanostomy tube system (TTS) has received FDA 510(k) clearance for use in children six months and older. The Hummingbird TTS is used in office-based pediatric ear tube procedures. The Maple Grove, Minnesota-based company designed the system to allow ENTs to make an incision and deliver, … [Read more...] about Preceptis Medical wins FDA clearance for expanded indications of Hummingbird tympanostomy tube system
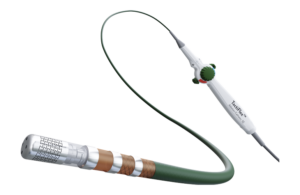
MedAlliance sirolimus-eluting balloon wins second FDA IDE
MedAlliance this week announced its Selution SLR sirolimus-eluting balloon received conditional FDA investigational device exemption (IDE) approval to begin a clinical trial for occlusive disease of the superficial femoral artery (SFA). The second IDE approval comes a few months after the Geneva, Switzerland-based company won IDE approval for … [Read more...] about MedAlliance sirolimus-eluting balloon wins second FDA IDE
BD issues Class I recall of some intraosseous needle set kits and drivers
BD (NYSE:BDX) has issued a class I recall of its intraosseous needle set kits for its intraosseous infusion system. BD issued the recall of its intraosseous needle set kits, manual driver kits and powered drivers due to three separate issues. The needle set kits stylets could be difficult to separate from the needle or not separate at all. The … [Read more...] about BD issues Class I recall of some intraosseous needle set kits and drivers
Freudenberg Medical launches HelixTC bioprocessing overmolded silicone ends
Freudenberg today launched its HelixTC silicone overmolded sanitary ends for bioprocessing fluid transfer applications. HelixTC is based on Freudenberg's PharmaFocus Premium product line of platinum-cured silicone tubing. It is an alternative to standard barbed TC connections. The new overmolded sanitary ends are available in standard or mini … [Read more...] about Freudenberg Medical launches HelixTC bioprocessing overmolded silicone ends
FDA clears Glaukos’ iStent Infinite
Glaukos this week announced it received FDA 510(k) clearance for its iStent Infinite trabecular micro-bypass system. The FDA clearance indication is for use in a standalone procedure to reduce elevated intraocular pressure (IOP) in patients who have primary open-angle glaucoma. San Clemente, California-based Glaukos designed iStent Infinite … [Read more...] about FDA clears Glaukos’ iStent Infinite
Abbott launches TactiFlex SE radiofrequency ablation catheter in Japan
Abbott this week announced it has launched its TactiFlex SE irrigation catheter in Japan. TactiFlex SE is a radiofrequency (RF) ablation catheter with a slit tip electrode with contact force technology. The tip of the catheter is flexible and conforms to the shape of the heart's wall for unique patient anatomies. The slit structure on the … [Read more...] about Abbott launches TactiFlex SE radiofrequency ablation catheter in Japan
Teleflex’s central venous access catheters awarded Vizient supply contract
Teleflex today announced it was awarded a contract for the supply of central venous access devices from Vizient, effective immediately. The agreement allows Vizient members to take advantage of special pricing for Teleflex's line of central venous access catheters (CVCs). The group purchasing agreement includes Teleflex's Arrowg+ard Blue Plus … [Read more...] about Teleflex’s central venous access catheters awarded Vizient supply contract