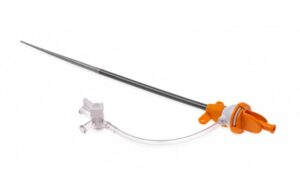
Abiomed (Nasdaq:ABMD) announced today that it received FDA 510(k) clearance for its Impella low-profile sheath. Danvers, Massachusetts-based Abiomed designed its sheath to maintain the same inner diameter as the existing 14 French (Fr) sheath used for Impella CP placement. However, the new sheath reduces the outer diameter by nearly 2 Fr. The … [Read more...] about FDA clears Impella low-profile sheath from Abiomed
B. Braun buys catheter securement device portfolio from Starboard
B. Braun Medical announced today that it acquired the Clik-FIX catheter securement device portfolio from Starboard Medical. Clik-FIX adds to B.Braun’s offerings that help improve patient experiences and outcomes for peripheral intravenous (IV) therapy. Get the full story at our sister site, MassDevice. … [Read more...] about B. Braun buys catheter securement device portfolio from Starboard
Teleflex launches new injectable midline catheter portfolio
Teleflex (NYSE:TFX) announced today that it released its new Arrow pressure injectable midline portfolio of catheters in North America. Wayne, Pennsylvania-based Teleflex designed its Arrow portfolio to help alleviate line identification confusion. The company said a lack of identification between midlines and catheters creates the potential for … [Read more...] about Teleflex launches new injectable midline catheter portfolio
FDA approves Medtronic drug-eluting stents for treating bifurcation lesions
Medtronic (NYSE:MDT) announced today that the FDA approved its Onyx drug-eluting stents (DES) for treating certain lesions. Approval covers the treatment of non-left main bifurcation lesions utilizing the provisional bifurcation stenting technique. The technique uses a single stent to treat the bifurcation in percutaneous coronary interventions … [Read more...] about FDA approves Medtronic drug-eluting stents for treating bifurcation lesions
Getinge partners with Medtronic to make Radiant balloon-expandable covered stent
Getinge today announced a partnership with Medtronic (NYSE:MDT) over the Radiant balloon-expandable stent, which received CE mark. Radiant is designed for use in chimney endovascular aneurysm repair (ChEVAR) with the Endurant II/IIs stent graft system. It maintains perfusion to renal arteries when used in combination with the stent graft. The … [Read more...] about Getinge partners with Medtronic to make Radiant balloon-expandable covered stent
Advanced NanoTherapies wins FDA breakthrough nod for drug-coated balloon
Advanced NanoTherapies announced today that it received FDA breakthrough device designation for its SirPlux Duo drug-coated balloon (DCB). Los Gatos, California-based Advanced NanoTherapies received the breakthrough nod for the use of the DCB in treating coronary artery disease in vessels less than 3.0mm. The regulatory win comes weeks after two … [Read more...] about Advanced NanoTherapies wins FDA breakthrough nod for drug-coated balloon
Merit Medical launches new Prelude Roadster guide sheath
Merit Medical Systems (Nasdaq:MMSI) announced today that it commercially released its Prelude Roadster guide sheath. South Jordan, Utah-based Merit designed its Prelude Roadster for deliverability, visibility, and resilience in tortuous peripheral vasculature anatomies. It helps to deliver devices used to diagnose and treat patients with … [Read more...] about Merit Medical launches new Prelude Roadster guide sheath
Penumbra launches RED reperfusion catheters in Europe
Penumbra (NYSE:PEN) announced today that it received the CE mark for its Penumbra RED reperfusion catheter and began the European launch. Alameda, California-based Penumbra designed its RED reperfusion catheter for the removal of all clot types in acute ischemic stroke (AIS) patients. The catheters are part of the Penumbra System — a fully … [Read more...] about Penumbra launches RED reperfusion catheters in Europe
J&J’s Biosense Webster launches mapping catheter for cardiac arrhythmias
Johnson & Johnson's Biosense Webster announced today that it launched the Octaray mapping catheter with TRUEref technology. Irvine, California-based Biosense Webster designed Octaray for mapping cardiac arrhythmias, including AFib. It is powered by the company's Carto 3 Version 7 system. The catheter has eight splines with improved … [Read more...] about J&J’s Biosense Webster launches mapping catheter for cardiac arrhythmias
Data backs Penumbra’s catheters for stroke care
Penumbra (NYSE:PEN) announced today that initial data support the use of its RED reperfusion catheters and the Penumbra System. Alameda, California-based Penumbra presented results from the initial Insight Registry for the RED reperfusion catheters and the sub-analyses of the Complete study for the Penumbra system at the 2022 World Federation of … [Read more...] about Data backs Penumbra’s catheters for stroke care