A string of positive news continues for Boston Scientific (NYSE: BSX), which this week announced FDA approval for its next-generation Watchman FLX. Last week, the company reported positive data for its Farapulse system, which led to a favorable reaction from analysts and the market. Just the week before, Boston Scientific received expanded FDA … [Read more...] about FDA approves next-gen Watchman FLX Pro from Boston Scientific
Catheters
Cordis names former Spectranetics CEO as executive chair
Interventional vascular tech company Cordis today announced that board member Scott Drake will be the new executive chair of its board of directors. Drake succeeds Duke Rohlen, who served as executive chair since Hellman & Friedman and KKR acquired the company from Cardinal Health in 2021. Rohlen will stay on the company's board of directors … [Read more...] about Cordis names former Spectranetics CEO as executive chair
ReCor Medical wins FDA panel nod for its renal denervation system
ReCor Medical announced that an FDA panel has voted in favor of the safety and efficacy of its Paradise ultrasound renal denervation (RDN) system for treating hypertension. On Aug. 22, the Circulatory Systems Devices Panel of the FDA's Medical Devices Advisory Committee voted 12–0 in favor with regard to safety and 8–3 in favor, with one vote … [Read more...] about ReCor Medical wins FDA panel nod for its renal denervation system
FDA fast-tracks aneurysm stabilization system from Nectero Medical
Nectero Medical announced today that the FDA granted fast track designation to its endovascular aneurysm stabilization treatment (EAST) system. Tempe, Arizona-based Nectero designed its EAST system to treat patients with abdominal aortic aneurysm (AAA). These AAAs have a maximum diameter of 3.5 cm to 5 cm. The company plans to initiate a … [Read more...] about FDA fast-tracks aneurysm stabilization system from Nectero Medical
Advanced NanoTherapies picks up $4M investment for drug-coated balloon development
Advanced NanoTherapies announced today that it collected a $4 million Series A extension from an undisclosed strategic investor. Los Gatos, California-based Advanced NanoTherapies received the investment from a medical device company. It also announced the successful treatment of the first cohort of study participants in its drug-coated balloon … [Read more...] about Advanced NanoTherapies picks up $4M investment for drug-coated balloon development
Teleflex extends Arrow ErgoPack system with hemodialysis, large bore catheters
Teleflex (NYSE:TFX) announced today that it released its Arrow ErgoPack complete kits with hemodialysis and large-bore catheters in the U.S. Wayne, Pennsylvania-based Teleflex launched this update to streamline insertion workflow for clinicians. The company also says it provides more options to suit their particular needs. Clinicians can now … [Read more...] about Teleflex extends Arrow ErgoPack system with hemodialysis, large bore catheters
J&J used RWE for expanded indications — and you can, too
Two J&J MedTech leaders shared advice to help medical device developers use real-world evidence (RWE) in FDA submissions. Real-world evidence (RWE) took a big step forward recently when the FDA approved expanded indications for Johnson & Johnson MedTech ablation catheters. For the first time, the federal medical device safety regulator … [Read more...] about J&J used RWE for expanded indications — and you can, too
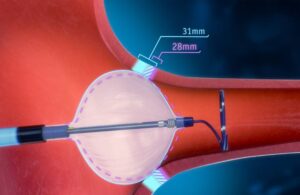
FDA approves POLARx cryoablation system from Boston Scientific
Boston Scientific [WtwhTicker symbol="BSX"](NYSE: BSX)[/WtwhTicker] announced today that it received FDA approval for its POLARx cryoablation system. The new system received an indication for the treatment of patients with paroxysmal AFib. It features the POLARx FIT cryoablation balloon catheter. This device enables two balloon sizes (28 mm and … [Read more...] about FDA approves POLARx cryoablation system from Boston Scientific
Acutus expects AcQBlate ablation catheter approval by end of 2023
Acutus Medical (Nasdaq:AFIB) reported positive second-quarter results, with some future plans that got analysts excited. Shares of AFIB rose 0.8% at 68¢ apiece in mid-afternoon trading today. MassDevice's MedTech 100 Index — which includes stocks of the world's largest medical device companies — fell 2.1%. BTIG analysts Marie Thibault and Sam … [Read more...] about Acutus expects AcQBlate ablation catheter approval by end of 2023
LeMaitre Vascular shares rise on 130% profit gains in Q2
LeMaitre Vascular (Nasdaq:LMAT) this week posted second-quarter results that beat the overall consensus on Wall Street. The Burlington, Massachusetts-based company reported profits of $8.1 million, or 36¢ per share, on sales of $50.1 million, for a sales growth of 19.02% and profit gains of 130.4% compared to Q3 2022. Earnings per share were … [Read more...] about LeMaitre Vascular shares rise on 130% profit gains in Q2