The medical device giant sponsored a retrospective claims analysis of a large, nationally representative insurance claims database. The results — recently published in Osteoporosis International — found that among the thousands of patients prescribed opioids, 48.7% stopped using after a BKP or VP procedure.
Another 8.4% reduced their prescription refills post-procedure.
The study was unique because it evaluated the impact of BKP and VP procedures on opioid prescription fills as a primary outcome, according to Dr. Joshua Hirsch, corresponding study author and practicing physician at Massachusetts General Hospital. Hirsch, in a Medtronic news release, also noted that the study used a more extensive, real-world study population versus previous studies.
“Given the current opioid epidemic and ongoing pain management crisis, we believe there is a need to better explore solutions that effectively address pain and support the elimination of oral opioids,” said Anu Codaty, VP and GM of Interventional Pain within Medtronic’s Neuromodulation business.
“Based on the findings of this study, there may be an added benefit for this patient population in reducing opioid-based harms,” Codaty said in a news release.
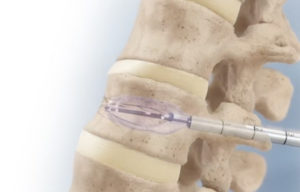
Medtronic’s Kyphon balloon kyphoplasty is a minimally invasive procedure in which cannulas smaller than drinking straws deliver balloons that lift fractured spine bone and return it to the correct position. A special cement fills the resulting cavity after balloon deflation.
The Kyphon V Premium vertebroplasty procedure uses a small-gauge osteo introducer. The introducer injects highly viscous bone cement into the trabeculae to stabilize a fracture.

