Medtronic's subsidiary Covidien issued a Class I recall of more than 1 million Palindrome and Mahurkar hemodialysis catheters. Covidien issued the recall due to a catheter hub defect that will connect both extension catheters. The defect could leak within the hub of a chronic dialysis catheter, which can lead to mixing of the arterial and venous … [Read more...] about Medtronic’s Covidien Palindrome and Mahurkar hemodialysis catheter recall is Class I
Acutus Medical wins regulatory approval in Japan for non-contact cardiac mapping catheter
Acutus Medical this week announced it received regulatory approval in Japan for its AcQMap high resolution imaging and mapping system and the AcQMap 3D imaging and mapping catheter. The approval represents a new era of non-contact arrhythmia management in Japan. Acutus Medical and its international alliance partner Biotronik have commenced … [Read more...] about Acutus Medical wins regulatory approval in Japan for non-contact cardiac mapping catheter
Ra Medical enters agreement to raise $6.2M, plans merger with Catheter Precision
Ra Medical Systems today announced a reduction in the exercise price of all existing warrants issued in February and has entered into a non-binding term sheet to merge with Catheter Precision. The price reduction affects all existing warrants issued in the February 2022 public offering, consisting of Series A warrants and Series B warrants, from … [Read more...] about Ra Medical enters agreement to raise $6.2M, plans merger with Catheter Precision
Light Line Medical enters licensing agreement with Mayo Clinic for catheter sterilization system
Light Line Medical today said it entered into a "know-how" license agreement with Mayo Clinic to advance its visible light infection prevention system for off-the-shelf catheters. The collaboration will focus on the technology in peritoneal dialysis catheters, Foley (urinary) catheters and endotracheal tubes and vascular catheters. It will also … [Read more...] about Light Line Medical enters licensing agreement with Mayo Clinic for catheter sterilization system
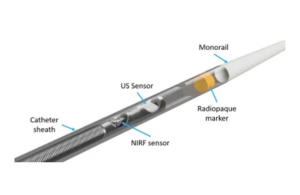
Intravascular Imaging launches coronary imaging catheter
Intravascular Imaging Incorporated this week launched its 3 Fr NIRF-IVUS imaging catheter for human coronary imaging. The catheter is designed in collaboration with the Massachusetts General Hospital an was tested ex-vivo in human coronary arteries and in vivo in preclinical studies. It uses intravascular ultrasound (IVUS) with near-infrared … [Read more...] about Intravascular Imaging launches coronary imaging catheter
Design challenges to overcome when developing cardiac ablation devices
It’s about figuring out how and where to go with the cardiac ablation and then engineering the best catheter-based delivery device, according to a top Acutus Medical scientist. Cardiac ablation is when a physician intentionally destroys a small amount of tissue in the heart to treat and prevent heart rhythm problems. The procedure creates … [Read more...] about Design challenges to overcome when developing cardiac ablation devices
Cerus Endovascular names Stephen Griffin as CEO
Cerus Endovascular today announced the appointment of Stephen Griffin to chief executive officer, effective immediately. Griffin has been with the company since 2014 and has served as president since 2018. He played a critical role in assembling the current Fremont, California-based company's team and helped establish the Contour brand … [Read more...] about Cerus Endovascular names Stephen Griffin as CEO
B. Braun launches Introcan Safety 2 IV catheters
B. Braun today announced that it launched its Introcan Safety 2 IV catheter with one-time blood control. Bethlehem, Pennsylvania-based B. Braun designed the Introcan Safety 2 catheter to reduce the risk of needle stick injuries and exposure to blood to make IV access safer for clinicians. The catheter is designed with a one-time blood control … [Read more...] about B. Braun launches Introcan Safety 2 IV catheters
FDA clears Ra Medical Dabra 2.0 catheter
Ra Medical Systems today announced its Dabra 2.0 catheter received FDA 510(k) clearance. The Carlsbad, California-based company designed the Dabra 2.0 catheter as part of its DABRA Excimer laser system. It has a braided over jacket design that improves deliverability and kink resistance when navigating tortuous anatomy. “While we are pleased … [Read more...] about FDA clears Ra Medical Dabra 2.0 catheter
Ancora Heart names Mark Miles as chief commercial officer
Ancora Heart today announced it appointed Mark Miles as chief commercial officer. Miles will oversee the Santa Clara, California-based company's commercialization strategy for the AccuCinch ventricular restoration system, a transcatheter procedure to treat enlarged left ventricles in the heart. “Mark’s track record in commercialization and … [Read more...] about Ancora Heart names Mark Miles as chief commercial officer