Ra Medical Systems today said it received a patent from the U.S. Patent and Trademark Office for a support catheter used with a small flexible liquid core catheter for laser ablation. The Carlsbad, California-based company was granted its 10th patent for the support catheter that is to be used in conjunction with the Dabra liquid core ablation … [Read more...] about Ra Medical Systems wins patent for laser ablation support catheter
CereVasc wins FDA IDE approval to begin eShunt pilot study
CereVasc today announced that it received an FDA investigational device exemption approval to begin a trial of the eShunt for normal pressure hydrocephalus. The Boston-based company can initiate its pilot trial of the eShunt system in patients with normal pressure hydrocephalus (NPH). The eShunt device offers a minimally invasive treatment with … [Read more...] about CereVasc wins FDA IDE approval to begin eShunt pilot study
Zeus launches multi-lumen tubing for steerable catheters
Zeus Industrial Products today announced that it has launched its PTFE Sub-Lite-Wall multi-lumen tubing to its product portfolio. Orangeburg, S.C.-based Zeus designed the multi-lumen tubing for steerable catheters. It has ultra-thin walls, certified USP Class VI biocompatibility, high structural integrity, improved planarity, high lubricity and … [Read more...] about Zeus launches multi-lumen tubing for steerable catheters
The top 10 catheter innovations of 2021
Last year was a big year for catheter innovation as medtech companies large and small received regulatory approvals for devices ranging from TAVR to single-use endoscopes. Catheter innovations weren't limited to just adults; Catheter-deployed devices for premature babies and toddlers were also approved and released. And catheter-based devices … [Read more...] about The top 10 catheter innovations of 2021
Hantel appoints VP of business development
Hantel recently announced that it appointed Ron Balogh as VP of business development. The Hayward, California-based medtech design and contract manufacturing company appointed Balogh on Jan. 3. He will be responsible for serving the company's current clients and growing the company by improving communications and its presence within the … [Read more...] about Hantel appoints VP of business development
Cook Medical wins FDA breakthrough designation for new drug-eluting stent
Cook Medical recently announced that it received FDA breakthrough designation for its drug-eluting stent for below the knee. Bloomington, Indiana-based Cook Medical designed the stent to treat patients who have chronic limb-threatening ischemia (CLTI). "CLTI is a debilitating disease of growing prevalence around the globe and this is Cook … [Read more...] about Cook Medical wins FDA breakthrough designation for new drug-eluting stent
FDA grants PMA for Cardiovascular Systems, OrbusNeich Scoreflex scoring balloon
Cardiovascular Systems and OrbusNeich this week announced that the OrbusNeich Scoreflex NC scoring percutaneous transluminal coronary angioplasty (PTCA) catheter has received FDA premarket approval. Scoreflex NC is a force PTCA scoring balloon with a dual-wire system that creates a focal stress pattern for safe and controlled plaque modification … [Read more...] about FDA grants PMA for Cardiovascular Systems, OrbusNeich Scoreflex scoring balloon
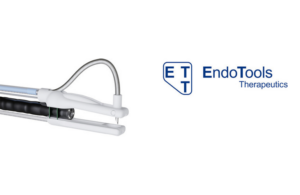
Endomina suturing tool wins FDA clearance
Endo Tools Therapeutics today announced that its Endomina suturing tool won FDA 510(k) clearance. The Belgium-based company designed the system for the endoscopic placement of sutures and approximation of soft tissue in the gastrointestinal tract in adults. Endomina has a universal triangulation platform and an instrument for tissue piercing … [Read more...] about Endomina suturing tool wins FDA clearance
Cardiovascular Systems’ recall of its Wirion embolic protection device is Class I
The FDA determined that Cardiovascular Systems’ recall of some of its Wirion embolic protection devices is Class I, the most serious kind. St. Paul, Minnesota–based Cardiovascular Systems is recalling all products and lots of its Wirion embolic protection device due to complaints of filter breakage during retrieval. It includes devices with … [Read more...] about Cardiovascular Systems’ recall of its Wirion embolic protection device is Class I
Medtronic Evolut Pro TAVR wins approval in China
Medtronic today announced that it received National Medical Products Administration approval in China for its CoreValve Evolut Pro TAVR system. The approval marks the first Medtronic TAVR system approved in China for patients with symptomatic severe aortic stenosis who are at high or extreme risk for open-heart surgery, according to the … [Read more...] about Medtronic Evolut Pro TAVR wins approval in China