Freudenberg Medical announced it is expanding its manufacturing operations in Alajuela, Costa Rica. The expansion project adds 8,600 ft² to the existing facility, including an additional ISO Class 7 cleanroom for catheter manufacturing and assembly, molding, extrusion and packaging, as well as added office space. Get the full story on our … [Read more...] about Freudenberg Medical expands Costa Rica operations
Catheters
HeMo Bioengineering aspiration catheter wins pre-market approval in China
HeMo Bioengineering this week announced that it received pre-market approval in China for its aspiration catheter. The Afentta intracranial thrombectomy aspiration catheter is a direct aspiration treatment option for acute ischemic stroke thrombectomy and has demonstrated lower post-stroke treatment disability rates and disease burden in … [Read more...] about HeMo Bioengineering aspiration catheter wins pre-market approval in China
Prytime Medical gains approvals for catheter
Prytime Medical Devices today said it received U.K. and CE mark approval for its ER-REBOA Plus catheter. The Boerne, Texas-based company designed the ER-REBOA (resuscitative endovascular balloon occlusion of the aorta) catheter to improve the ease of use for controlling non-compressible truncal hemorrhage. "With this milestone, the new and … [Read more...] about Prytime Medical gains approvals for catheter
Boston Scientific has a serious stent recall problem
The FDA announced today that it deemed Boston Scientific’s recall of its VICI SDS and VICI RDS venous stent systems as Class I, the most serious kind of recall. Marlborough, Mass.-based Boston Scientific’s VICI SDS and VICI RDS venous stent systems treat obstructions and occlusions in narrowed or blocked venous veins. Get the full story at our … [Read more...] about Boston Scientific has a serious stent recall problem
Is renal denervation back as a high blood pressure treatment?
Medtronic (NYSE:MDT) today reported clinically significant and sustained blood pressure reduction among nearly 3,000 people with uncontrolled hypertension treated with the company’s Symplicity renal denervation system. The study results, reported today out of the 2021 EuroPCR Annual Meeting, come the same week as ReCor Medical touted its … [Read more...] about Is renal denervation back as a high blood pressure treatment?
ReCor Medical has positive renal denervation study news
ReCor Medical‘s renal denervation significantly reduced blood pressure among people with drug-resistant hypertension, according to a new study reported at the American College of Cardiology’s 70th Annual Scientific Session. The study news, announced yesterday, could suggest a turnaround in fortunes for renal denervation technology. One … [Read more...] about ReCor Medical has positive renal denervation study news
Edwards Lifesciences tricuspid valve reduced leakage in 98% of patients
Edwards Lifesciences‘s Evoque tricuspid valve replacement system reduces tricuspid heart valve leakage to non-traceable or mild leakage in 98% of patients, according to a new study reported at the American College of Cardiology’s 70th Annual Scientific Session. Evoque is designed to replace native tricuspid valves without open-heart surgery. The … [Read more...] about Edwards Lifesciences tricuspid valve reduced leakage in 98% of patients
Galaxy Medical, Japan Lifeline ink distribution agreement for ablation catheters
Galaxy Medical and Japan Lifeline today said they signed an exclusive distribution agreement for the Alpha1 ablation catheter. Under the agreement Galaxy will pursue U.S. regulatory approval for Japan Lifeline's Alpha1 in support of the commercialization of Galaxy's Centauri pulsed electric field system, which can be used with the ablation … [Read more...] about Galaxy Medical, Japan Lifeline ink distribution agreement for ablation catheters
BD enrolls first patients in arteriovenous endovascular fistula post-market trial
BD (NYSE: BDX) recently announced that it has begun enrollment for its post-market surveillance study of its WavelinQ arteriovenous endovascular fistula device. The Connect-AV trial is a prospective, single-arm, open-label study that will follow WavelinQ EndoAVF system patients for 24 months. Its dual primary effectiveness endpoints are the … [Read more...] about BD enrolls first patients in arteriovenous endovascular fistula post-market trial
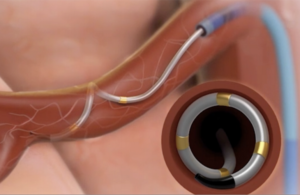
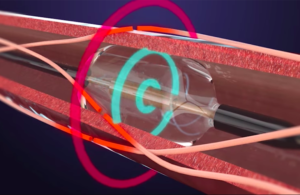
Boston Scientific initiates coronary drug-coated balloon study in U.S.
Boston Scientific (NYSE:BSX) this week launched its Agent IDE trial for its Agent drug-coated balloon. The U.S. prospective, randomized clinical trial will evaluate the safety and effectiveness of a drug-coated balloon (DCB) in patients with coronary in-stent restenosis in lesions up to 26 mm in length in a coronary artery 2.0 mm to 4.0 mm in … [Read more...] about Boston Scientific initiates coronary drug-coated balloon study in U.S.