Front Line Medical Technologies today announced that its COBRA-OS bleeding control device won FDA 510(k) clearance. The COBRA-OS (control of bleeding, resuscitation, arterial occlusion system) is a 4 French REBOA (resuscitative endovascular balloon occlusion of the aorta) device that provides full occlusion, intermittent occlusion or partial … [Read more...] about FDA clears Front Line Medical arterial occlusion system
Catheters
FDA grants PMA to Abiomed’s next-generation Impella RP
Abiomed (NSDQ:ABMD) announced today that the FDA granted premarket approval (PMA) to its Impella RP with SmartAssist. Danvers, Mass.–based Abiomed received PMA for the Impella RP right heart pump with SmartAssist as a safe and effective treatment for acute right heart failure for up to 14 days, according to a news release. Get the full story at … [Read more...] about FDA grants PMA to Abiomed’s next-generation Impella RP
CardioFocus names new CFO
CardioFocus today said it appointed Stephan Ogilvie as chief financial officer. Prior to joining the Marlborough, Mass.-based company, Ogilvie served as managing director of healthcare investment banking at B Riley Securities. He has also served as VP of corporate development at Nuvasive and has held numerous leadership roles at ThinkEquity … [Read more...] about CardioFocus names new CFO
Stratus Medical receives CE mark for Nimbus RF multitined expandable electrode, Vesta RF cannula
Stratus Medical this week said it received CE mark approval for its Nimbus RF multitined expandable electrode and Vesta RF cannula. The Nimbus multitined expandable electrode is designed to create a large 8 mm to 10 mm prolate spheroid lesion for pain relief. More than 100,000 of the devices have been used to treat chronic pain in patients in … [Read more...] about Stratus Medical receives CE mark for Nimbus RF multitined expandable electrode, Vesta RF cannula
FDA clears Penumbra’s Red 62 reperfusion catheter for removing brain blood clots
Penumbra (NYSE:PEN) today said its Red 62 reperfusion catheter has received FDA 510(k) clearance. Alameda, Calif.-based Penumbra designed the catheter to navigate complex distal vessel anatomy and deliver powerful aspiration with its Penumbra Engine to remove blood clots in acute ischemic stroke patients with large vessel occlusions. “In my … [Read more...] about FDA clears Penumbra’s Red 62 reperfusion catheter for removing brain blood clots
CardioFocus wins Japan regulatory approval for HeartLight X3 system
CardioFocus today said its HeartLight X3 catheter ablation technology received approval from the Japanese Ministry of Health, Labor and Welfare. Marlborough, Mass.-based CardioFocus designed the catheter ablation technology for controlled and consistent pulmonary vein isolation to treat atrial fibrillation (AFib). "MHLW's regulatory approval … [Read more...] about CardioFocus wins Japan regulatory approval for HeartLight X3 system
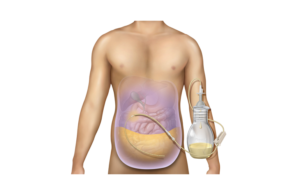
BD home peritoneal catheter system wins expanded FDA clearance
BD today said it has received FDA 510(k) clearance for its PeritX peritoneal catheter system. Franklin Lakes, N.J.-based BD designed the catheter system to drain symptomatic, recurrent non-malignant ascites, which causes a build-up of fluid in the abdomen. The company's PleurX peritoneal catheter system was first introduced in 2005 to drain … [Read more...] about BD home peritoneal catheter system wins expanded FDA clearance
Medtronic wins expanded FDA approval for cryoablation catheters
Medtronic (NYSE:MDT) announced today that it received FDA expanded approval for its Arctic Front family of cardiac cryoablation catheters. Fridley, Minn.-based Medtronic's Arctic Front family of catheters treat recurrent symptomatic paroxysmal atrial fibrillation (AFib) as an alternative to antiarrhythmic drug (AAD) therapy as an initial rhythm … [Read more...] about Medtronic wins expanded FDA approval for cryoablation catheters
Midwest Interventional Systems completes funding round
Midwest Interventional Systems (MIS) recently announced the completion of a Series A financing with a group of private healthcare investors. Oversubscribed from original expectations, the Series A round will enable MIS to accelerate the vertical integration of capabilities and continue its growth trajectory and support expansion initiatives that … [Read more...] about Midwest Interventional Systems completes funding round
How Abbott engineered a catheter-delivered device for premature babies
Abbott’s experience designing its Piccolo device provides essential lessons for medical device creators who want to spark a pediatric medical device renaissance. The Amplatzer Piccolo occluder from Abbott is one of the first medical devices that health providers can implant in premature babies weighing as little as 2 lb to treat patent ductus … [Read more...] about How Abbott engineered a catheter-delivered device for premature babies