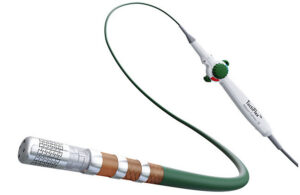
For Abbott, combining two innovations into one AFib-treating system required manufacturing fine-tuning. The project’s R&D director explains. Pulsed field ablation (PFA) has generated a great deal of buzz for its potential to reduce complications in procedures for treating atrial fibrillation (AFib). But even as Abbott is developing its Volt … [Read more...] about How Abbott created its next-gen RF ablation system
Product Development
Microbot Medical expands U.S. operations to prep for first in-human clinical study
Microbot Medical, the developer of the Liberty endovascular robotic surgical system, is building on the momentum from the positive results of its good laboratory practices (GLP) pre-clinical study. The company is adding a clinical research associate to support its pending investigational device exemption, or IDE, submission to commence its … [Read more...] about Microbot Medical expands U.S. operations to prep for first in-human clinical study
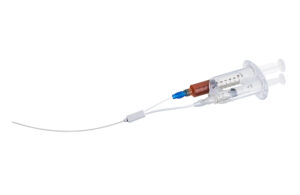
VVT Medical announces first commercial ScleroSafe case in U.S.
VVT Medical recently announced the successful completion of the first commercial U.S. varicose vein treatment using its ScleroSafe platform. Dr. Steve Elias performed the procedure at the Center for Vein Disease, part of the Englewood Health Network in Englewood, New Jersey. “The Sclerosafe procedure takes less than 30 minutes, requires only … [Read more...] about VVT Medical announces first commercial ScleroSafe case in U.S.
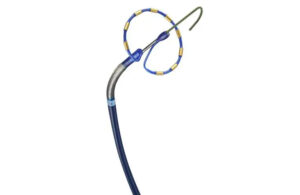
Medtronic pulsed-field ablation system wins CE mark
Medtronic said today that it's received a CE mark for its PulseSelect pulsed field ablation system. In addition, the medtech giant received European Union approval for its Nitron CryoConsole, which builds upon the legacy of the company's cryo franchise with features to optimize the workflow for cryoballoon ablation. The news of the regulatory … [Read more...] about Medtronic pulsed-field ablation system wins CE mark
Two-armed surgical robot prototype targets pediatric brain tumors
Researchers say they've developed a surgical robot for removing brain tumors in children, and that it could also offer a less invasive, safer option for adult neurosurgery and other procedures. The trick is using hollow, nitinol robot arms to allow neurosurgeons to swap tools during a tumor resection procedure, said Pierre DuPont. He's the chief … [Read more...] about Two-armed surgical robot prototype targets pediatric brain tumors
After recall and relaunch, Medtronic wants to go global with its catheter-delivered Harmony valve
Each Medtronic Harmony valve is sewn by hand to attach laser-cut pig tissue to the nitinol that makes this minimally invasive heart implant possible. Medtronic’s Harmony transcatheter pulmonary valve (TPV) design is paying off after engineers solved a delivery catheter recall and relaunched the system this year. The Harmony TPV uses pig tissue, … [Read more...] about After recall and relaunch, Medtronic wants to go global with its catheter-delivered Harmony valve
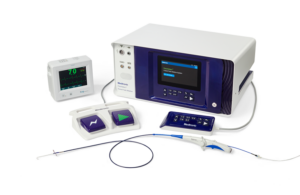
Medtronic wins CE mark for Affera ablation system
Medtronic (NYSE:MDT) announced today that it received CE mark approval for its Affera mapping and ablation system. The Affera system includes the Sphere-9 catheter and Affera Prism-1 mapping software. Together, the system integrates Sphere-9 pulsed-field ablation (PFA), radiofrequency (RF) and high-density (HD) mapping. It maps and ablates … [Read more...] about Medtronic wins CE mark for Affera ablation system
The biggest cardiology tech stories from ACC.23
Major medtech players presented a range of intriguing studies at a gathering of some of the biggest names in the cardiology tech space. Some of the hottest topics were covered this past week at the American College of Cardiology’s Annual Scientific Session Together With the World Congress of Cardiology (ACC.23/WCC) in New Orleans. Ablation, … [Read more...] about The biggest cardiology tech stories from ACC.23
Medtronic pulsed-field ablation system exceeds safety goal in study
Medtronic (NYSE:MDT) announced today that its PulseSelect pulsed-field ablation (PFA) system exceeded its safety performance goal in a clinical trial. PulseSelect registered an adverse event rate of 0.7%. Medtronic said that marks one of the lowest adverse event rates of any previous FDA investigational device exemption (IDE) trial for AFib … [Read more...] about Medtronic pulsed-field ablation system exceeds safety goal in study
Alleviant Medical raises $75M for no-implant heart failure treatment
Alleviant Medical has raised $75 million to support a global pivotal trial of its catheter-delivered interatrial shunt to treat heart failure. The FDA provided the Austin, Texas–based company with an Investigational Device Exemption for the clinical trial in November 2022. Researchers have designed the ALLAY-HF study to demonstrate the safety … [Read more...] about Alleviant Medical raises $75M for no-implant heart failure treatment