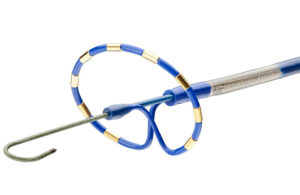
Biosense Webster’s pulsed-field ablation (PFA) catheter design and development starts in the heart.
That’s where PFA treats atrial fibrillation (AFib) by killing cardiac cells to isolate errant heart signals and prevent them from triggering irregular heartbeats. PFA uses electric fields to permanently open holes in the walls of heart cells … [Read more...] about High voltage in the heart: PFA catheter design tips from Biosense Webster
Uncategorized
High voltage in the heart: PFA catheter design tips from Biosense Webster
What’s so special about pulsed field ablation? Medtronic EVP Sean Salmon explains
Medtronic’s PulseSelect Pulsed Field Ablation (PFA) System — which won the first FDA approval for PFA to treat atrial fibrillation (AFib) — is just the start of a wave of new PFA devices expected to hit the market. Medtronic is lining up another PFA cardiac ablation system for approval, while competitor Boston Scientific anticipates approval of … [Read more...] about What’s so special about pulsed field ablation? Medtronic EVP Sean Salmon explains
Cerenovus President Mark Dickinson on the future of stroke care
Cerenovus President Mark Dickinson forecasts the innovative technologies that will advance stroke care in the coming years. It's getting harder to beat aspiration systems for fast and simple thrombectomies to remove blood clots that are blocking oxygen from a stroke patient's brain. That's according to Cerenovus Worldwide President Mark … [Read more...] about Cerenovus President Mark Dickinson on the future of stroke care
Boston Scientific’s Dr. Michael Jaff on the future of peripheral intervention
Advanced technologies have the potential to take peripheral therapy "to a different stratosphere," says Dr. Michael Jaff, the chief medical officer and VP of the Boston Scientific Peripheral Interventions business. He characterized them as similar to the ability to combine devices with drugs, which he called the biggest transformational change … [Read more...] about Boston Scientific’s Dr. Michael Jaff on the future of peripheral intervention
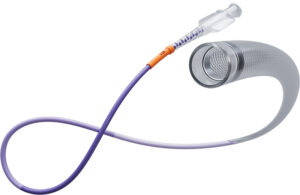
How LimFlow’s foot-saving system prevents amputations in patients with no other options
LimFlow’s breakthrough system for treating chronic limb-threatening ischemia (CLTI) is the first of its kind approved by the FDA for this severe form of peripheral artery disease (PAD). For CLTI patients who have lost blood flow below their knee and have no other suitable endovascular or surgical treatment options available, the LimFlow System … [Read more...] about How LimFlow’s foot-saving system prevents amputations in patients with no other options
Two-armed surgical robot prototype targets pediatric brain tumors
Researchers say they've developed a surgical robot for removing brain tumors in children, and that it could also offer a less invasive, safer option for adult neurosurgery and other procedures. The trick is using hollow, nitinol robot arms to allow neurosurgeons to swap tools during a tumor resection procedure, said Pierre DuPont. He's the chief … [Read more...] about Two-armed surgical robot prototype targets pediatric brain tumors
Zeus opens Minnesota catheter manufacturing facility
Zeus has opened its catheter manufacturing facility in Arden Hills, Minnesota, celebrating the new facility with a grand opening. The 75,600-ft² building is home to CathX Medical, which Zeus acquired 2021. The facility has advanced R&D lab and cleanroom space for designing, developing and validating medical catheter prototypes. The … [Read more...] about Zeus opens Minnesota catheter manufacturing facility
Medical nitinol processing: How NiTi is turned into wire, tubes and sheets for medical devices
Medical nitinol processing transforms raw nickel-titanium alloy into wire, coils, tubes and sheets for medical and dental device manufacturers. We previously covered nitinol's journey from the earth's crust through high-temperature melting crucibles and forging operations to create nitinol ingots and then smaller, more workable shapes such as … [Read more...] about Medical nitinol processing: How NiTi is turned into wire, tubes and sheets for medical devices
J&J used RWE for expanded indications — and you can, too
Two J&J MedTech leaders shared advice to help medical device developers use real-world evidence (RWE) in FDA submissions. Real-world evidence (RWE) took a big step forward recently when the FDA approved expanded indications for Johnson & Johnson MedTech ablation catheters. For the first time, the federal medical device safety regulator … [Read more...] about J&J used RWE for expanded indications — and you can, too
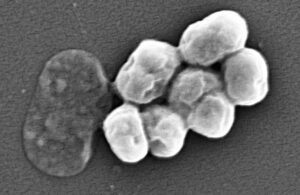
Some hospitalized patients’ infections may develop from their own bacteria in catheters
Researchers in Missouri discovered evidence of infections forming from hospitalized patients' own bacteria in catheters. Studying mice, researchers at Washington University School of Medicine in St. Louis discovered that urinary tract infections (UTIs) can arise after sterile catheter tube insertion into the urinary tract. This can occur even … [Read more...] about Some hospitalized patients’ infections may develop from their own bacteria in catheters