NanoVibronix today received publication of guidance from National Institute for Health and Care Excellence (NICE) in the U.K. for its UroShield device. England's Department of Health and Social Care's NICE issued guidance suggesting that UroShield has the potential to provide significant patient and healthcare system benefits to prevent … [Read more...] about NanoVibronix’s UroShield receives positive guidance from UK’s NICE
Catheters
Ra Medical Systems secures U.S. patent for new laser ablation catheters
Ra Medical Systems today announced that it received a patent from the U.S. Patent and Trademark Office for its liquid-filled laser ablation catheters with expanded distal optical windows. The patent describes a catheter with an increased ablation surface area at the distal catheter tip. It is the 11th U.S. patent issued to the Carlsbad, … [Read more...] about Ra Medical Systems secures U.S. patent for new laser ablation catheters
Interrad Medical’s SecurAcath awarded Innovative Technology contract by Vizient
Interrad Medical today announced that it received an Innovative Technology contract from Vizient for its SecurAcath technology. The Innovative Technology program by Vizient identifies medical devices that can improve clinical care and enhance the business model for healthcare organizations. It gives device companies visibility in Vizient's … [Read more...] about Interrad Medical’s SecurAcath awarded Innovative Technology contract by Vizient
Medtronic issues voluntary recall for certain In.Pact drug-coated balloon catheters
Medtronic (NYSE:MDT) announced today that it voluntarily recalled a subset of its In.Pact Admiral and In.Pact AV balloon catheters. The company initiated the recent recall of the paclitaxel-coated percutaneous transluminal angioplasty (PTA) balloon catheters due to the potential for pouch damage resulting in a loss of sterility. Get the full … [Read more...] about Medtronic issues voluntary recall for certain In.Pact drug-coated balloon catheters
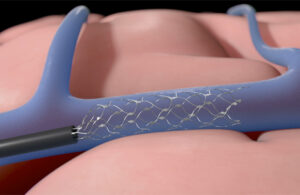
Cook Medical wins FDA breakthrough designation for Thoraco+ endovascular system
Cook Medical today announced that its Zenith Thoraco+ endovascular system received FDA breakthrough device designation. Thoraco+ is the company's next-generation endovascular graft that is indicated for the endovascular treatment of patients with thoracoabdominal aortic aneurysms. “We are excited to receive an FDA breakthrough device … [Read more...] about Cook Medical wins FDA breakthrough designation for Thoraco+ endovascular system
FDA approves Edwards Lifesciences’ tissue valve replacement for heart’s mitral position
Edwards Lifesciences (NYSE:EW) announced today that the FDA approved its Mitris Resilia tissue valve replacement system. Irvine, California-based Edwards designed the Mitris Resilia valve with a saddle-shaped sewing cuff that mimics the asymmetric shape of the native mitral valve. It features a low-profile frame for avoiding obstruction of the … [Read more...] about FDA approves Edwards Lifesciences’ tissue valve replacement for heart’s mitral position
4C Medical closes $35M Series C financing round, appoints CEO
4C Medical Technologies today announced that it completed a $35.4 million Series C financing round and appointed Saravana Kumar as president and CEO. The preferred stock financing round was led by MicroPort CardioFlow Medtech. As part of the closing, Kumar was appointed to lead the company after having served as interim president and CEO since … [Read more...] about 4C Medical closes $35M Series C financing round, appoints CEO
Synchron says study shows safety of its brain-computer interface implant
Four ALS patients with a Synchron Stentrode brain implant had no serious adverse events one year after their procedure, which allowed the paralyzed patients to control a computer for online shopping, banking and text communication without using their hands or voice for input. New York-based Synchron said this study demonstrated the safety of its … [Read more...] about Synchron says study shows safety of its brain-computer interface implant
Tractus Vascular announces first-in-human use of Crossing Support Catheter
Tractus Vascular recently announced the first-in-human use of its Tractus Crossing Support Catheter. Drs. John Rundback and Kevin Herman at American Endovascular and Amputation Prevention in West Orange N.J. performed the first procedures using Tractus CSC to treat patients with challenging cases of critical limb ischemia such as chronic total … [Read more...] about Tractus Vascular announces first-in-human use of Crossing Support Catheter
Endovastec Hyperflex balloon catheter wins marketing nod in Japan
Endovastec today announced it received registration approval from Japan Pharmaceuticals and Medical Devices Agency for its Hyperflex balloon catheter. The Shanghai-based heart device maker designed Hyperflex to assist in the dilation of aortic stent graft. It is the company's first device approved for marketing in Japan. Hyperflex previously … [Read more...] about Endovastec Hyperflex balloon catheter wins marketing nod in Japan