Boston Scientific (NYSE:BSX) today announced the acquisition of Obsidio and its blood vessel embolization material technology. The financial details of the deal were not disclosed. Obsidio develops gel embolic material (GEM) technology that is used for the embolization of blood vessels in the peripheral vasculature. It is a semi-solid, gel-link … [Read more...] about Boston Scientific acquires Obsidio and its gel embolic material
Catheters
MedAlliance sirolimus-eluting balloon wins second FDA IDE
MedAlliance this week announced its Selution SLR sirolimus-eluting balloon received conditional FDA investigational device exemption (IDE) approval to begin a clinical trial for occlusive disease of the superficial femoral artery (SFA). The second IDE approval comes a few months after the Geneva, Switzerland-based company won IDE approval for … [Read more...] about MedAlliance sirolimus-eluting balloon wins second FDA IDE
FDA clears Glaukos’ iStent Infinite
Glaukos this week announced it received FDA 510(k) clearance for its iStent Infinite trabecular micro-bypass system. The FDA clearance indication is for use in a standalone procedure to reduce elevated intraocular pressure (IOP) in patients who have primary open-angle glaucoma. San Clemente, California-based Glaukos designed iStent Infinite … [Read more...] about FDA clears Glaukos’ iStent Infinite
Abbott launches TactiFlex SE radiofrequency ablation catheter in Japan
Abbott this week announced it has launched its TactiFlex SE irrigation catheter in Japan. TactiFlex SE is a radiofrequency (RF) ablation catheter with a slit tip electrode with contact force technology. The tip of the catheter is flexible and conforms to the shape of the heart's wall for unique patient anatomies. The slit structure on the … [Read more...] about Abbott launches TactiFlex SE radiofrequency ablation catheter in Japan
Teleflex’s central venous access catheters awarded Vizient supply contract
Teleflex today announced it was awarded a contract for the supply of central venous access devices from Vizient, effective immediately. The agreement allows Vizient members to take advantage of special pricing for Teleflex's line of central venous access catheters (CVCs). The group purchasing agreement includes Teleflex's Arrowg+ard Blue Plus … [Read more...] about Teleflex’s central venous access catheters awarded Vizient supply contract
Medtronic’s Covidien Palindrome and Mahurkar hemodialysis catheter recall is Class I
Medtronic's subsidiary Covidien issued a Class I recall of more than 1 million Palindrome and Mahurkar hemodialysis catheters. Covidien issued the recall due to a catheter hub defect that will connect both extension catheters. The defect could leak within the hub of a chronic dialysis catheter, which can lead to mixing of the arterial and venous … [Read more...] about Medtronic’s Covidien Palindrome and Mahurkar hemodialysis catheter recall is Class I
Acutus Medical wins regulatory approval in Japan for non-contact cardiac mapping catheter
Acutus Medical this week announced it received regulatory approval in Japan for its AcQMap high resolution imaging and mapping system and the AcQMap 3D imaging and mapping catheter. The approval represents a new era of non-contact arrhythmia management in Japan. Acutus Medical and its international alliance partner Biotronik have commenced … [Read more...] about Acutus Medical wins regulatory approval in Japan for non-contact cardiac mapping catheter
Ra Medical enters agreement to raise $6.2M, plans merger with Catheter Precision
Ra Medical Systems today announced a reduction in the exercise price of all existing warrants issued in February and has entered into a non-binding term sheet to merge with Catheter Precision. The price reduction affects all existing warrants issued in the February 2022 public offering, consisting of Series A warrants and Series B warrants, from … [Read more...] about Ra Medical enters agreement to raise $6.2M, plans merger with Catheter Precision
Light Line Medical enters licensing agreement with Mayo Clinic for catheter sterilization system
Light Line Medical today said it entered into a "know-how" license agreement with Mayo Clinic to advance its visible light infection prevention system for off-the-shelf catheters. The collaboration will focus on the technology in peritoneal dialysis catheters, Foley (urinary) catheters and endotracheal tubes and vascular catheters. It will also … [Read more...] about Light Line Medical enters licensing agreement with Mayo Clinic for catheter sterilization system
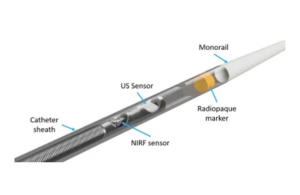
Intravascular Imaging launches coronary imaging catheter
Intravascular Imaging Incorporated this week launched its 3 Fr NIRF-IVUS imaging catheter for human coronary imaging. The catheter is designed in collaboration with the Massachusetts General Hospital an was tested ex-vivo in human coronary arteries and in vivo in preclinical studies. It uses intravascular ultrasound (IVUS) with near-infrared … [Read more...] about Intravascular Imaging launches coronary imaging catheter