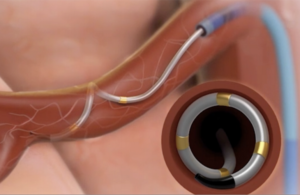
Medtronic (NYSE:MDT) will continue the clinical study of its Symplicity Spyral renal denervation (RDN) system for hypertension into next year after lacking the positive results needed to end enrollment early. Fridley, Minnesota-based Medtronic said last month that it hoped to present results of the Spyral HTN-ON MED trial at the Cardiovascular … [Read more...] about Medtronic faces longer road to renal denervation
Study finds single-use bronchoscopes reduce hospital readmission rates by half
A study of 14,228 bronchoscopy procedures found sterile, disposable scopes reduced readmission rates by more than half. The study, authored by Ambu consultant Dr. Hudson Garrett, analyzed procedures at inpatient hospitals as well as outpatient ambulatory facilities. He found that 3.6 percent of patients treated with a single-use, flexible … [Read more...] about Study finds single-use bronchoscopes reduce hospital readmission rates by half
OneProjects raises $17M to advance 4D cardiac imaging
Irish-German cardiac imaging startup OneProjects today announced $8 million in Series A2 funding and two grants totaling $9 million. The company will use the new funds to complete product development, conduct clinical trials, gain FDA clearance and prepare for U.S. commercialization of the first product developed from its proprietary 4D cardiac … [Read more...] about OneProjects raises $17M to advance 4D cardiac imaging
Raumedic plans U.S. headquarters expansion
Raumedic today said it intends to expand its clean room manufacturing facilities at its U.S. headquarters in Mills River, North Carolina. The new clean room is expected to be completed by February 2022 and used for new product lines. The Helmbrechts, Germany-based medical device component developer and manufacturer built its existing … [Read more...] about Raumedic plans U.S. headquarters expansion
How a future-minded medtech engineer finds inspiration, innovation and creativity
The future of medtech is the focus of TE Connectivity’s Advanced Technology Group, a team of experienced engineers tasked with identifying trends and developing new technology for devices and challenges that might not yet even exist. Marie Ottum, principal R&D engineer in TE Connectivity’s Advanced Technology Group and global leader of the … [Read more...] about How a future-minded medtech engineer finds inspiration, innovation and creativity
InTRAvent wins FDA 510(k) clearance for Solopass bedside neuro-navigation device
InTRAvent has announced FDA 510(k) clearance of its Solopass system for assisting frontal placement of intraventricular catheters deep within the brain. The Hershey, Pennsylvania-based company said yesterday that it designed the device with 2D and 3D ultrasound imaging, artificial intelligence and trajectory guidance for intraprocedural … [Read more...] about InTRAvent wins FDA 510(k) clearance for Solopass bedside neuro-navigation device
Medtronic releases Navablate study results for bronchoscopic microwave ablation
Medtronic (NYSE:MDT) reported new clinical data from its Navablate study of bronchoscopic microwave ablation with its Emprint ablation catheter kit to treat malignant lung nodules. “Patients with malignant lesions in the lung often have limited therapy options due to lesion locations, comorbidities and treatment side effects,” said one of the … [Read more...] about Medtronic releases Navablate study results for bronchoscopic microwave ablation
Putnam Plastics expands HQ with new tubing manufacturing facility
Medical extrusion firm Putnam Plastics Corp. has completed the construction of a 57,000-square-foot addition to its headquarters in northeast Connecticut. Dayville, Conn.–based Putnam, which makes advanced extrusions and assemblies for minimally invasive medical devices, will have new extrusion lines in the clean manufacturing space as well as … [Read more...] about Putnam Plastics expands HQ with new tubing manufacturing facility