Medical nitinol processing transforms raw nickel-titanium alloy into wire, coils, tubes and sheets for medical and dental device manufacturers. We previously covered nitinol's journey from the earth's crust through high-temperature melting crucibles and forging operations to create nitinol ingots and then smaller, more workable shapes such as … [Read more...] about Medical nitinol processing: How NiTi is turned into wire, tubes and sheets for medical devices
J&J used RWE for expanded indications — and you can, too
Two J&J MedTech leaders shared advice to help medical device developers use real-world evidence (RWE) in FDA submissions. Real-world evidence (RWE) took a big step forward recently when the FDA approved expanded indications for Johnson & Johnson MedTech ablation catheters. For the first time, the federal medical device safety regulator … [Read more...] about J&J used RWE for expanded indications — and you can, too
After recall and relaunch, Medtronic wants to go global with its catheter-delivered Harmony valve
Each Medtronic Harmony valve is sewn by hand to attach laser-cut pig tissue to the nitinol that makes this minimally invasive heart implant possible. Medtronic’s Harmony transcatheter pulmonary valve (TPV) design is paying off after engineers solved a delivery catheter recall and relaunched the system this year. The Harmony TPV uses pig tissue, … [Read more...] about After recall and relaunch, Medtronic wants to go global with its catheter-delivered Harmony valve
Zeus nears completion of Minnesota catheter manufacturing facility
NEWS RELEASE: Medical device manufacturing gets major boost as Zeus opens state-of-the-art facility Zeus, a global leader in advanced polymer solutions, announced today that construction is almost complete on a state-of-the-art catheter manufacturing facility in Arden Hills, Minnesota. The location, scale, and on-site capabilities across R&D, … [Read more...] about Zeus nears completion of Minnesota catheter manufacturing facility
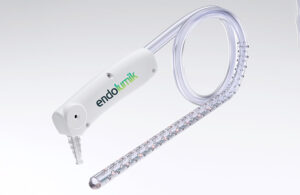
Endolumik’s illuminated device takes a big step for safety
Endolumik's story started a few years ago as the FDA sounded the alarm over the risks of internal surgical staplers. In 2019, the federal agency warned healthcare providers that it had received more than 41,000 medical device reports (MDRs) related to surgical staplers and staples for internal use from 2011 to 2018. Those MDRs tallied more than … [Read more...] about Endolumik’s illuminated device takes a big step for safety
Junkosha names new US leadership
Japanese fluoropolymer supplier Junkosha today announced new leadership for its U.S. subsidiary. Joe Rowan has retired as CEO and president of Junkosha USA but will remain as an advisor as Mike Winterling takes over as chief operating officer. “Our aim is to be acknowledged as the most responsive partner for specialized high-performance … [Read more...] about Junkosha names new US leadership
How Penumbra’s smart-sucking algorithms and catheters speed up clot removal
Blood loss is a big problem when using aspiration catheters to remove blood clots from patients' veins and arteries. "You suck out the clot, you also suck out the blood," said Sandra Lesenfants, president of the interventional business at Penumbra (NYSE:PEN). Algorithms developed by Penumbra's software team for the company's continuous … [Read more...] about How Penumbra’s smart-sucking algorithms and catheters speed up clot removal
Freudenberg Medical expands medical tubing production to Massachusetts
NEWS RELEASE: Freudenberg Medical Expands Medical Tubing Production to Massachusetts Freudenberg Medical, a global contract design and manufacturing provider to the medical device and pharmaceutical industry, has added custom silicone extrusion and medical and biopharma tube processing to its newest cleanroom manufacturing operation and global … [Read more...] about Freudenberg Medical expands medical tubing production to Massachusetts
FDA hits Olympus with warning letters after factory inspections
The FDA today released warning letters against Olympus Medical Systems Corp. and Olympus Corp. subsidiary Aizu Olympus following inspections of their endoscope and reprocessor manufacturing facilities. The warning letters allege medical device reporting (MDR) and quality system violations at the manufacturing operations. “Olympus’ highest … [Read more...] about FDA hits Olympus with warning letters after factory inspections
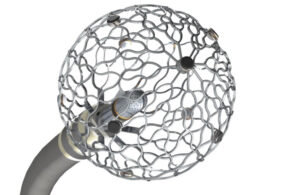
Why Affera’s cardiac ablation technology is worth $1B to Medtronic
Affera started in 2014 with a simple goal that paid off when Medtronic (NYSE:MDT) bought the company for up to $1 billion this year. Achieving that goal, however, took some unconventional and sometimes difficult design choices, Affera founder and CEO Doron Harlev said. Newton, Massachusetts–based Affera's system diagnoses, maps and treats … [Read more...] about Why Affera’s cardiac ablation technology is worth $1B to Medtronic