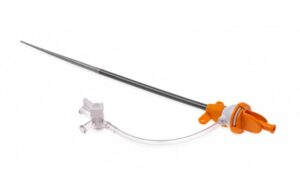
MedAlliance today announced it received FDA investigational device exemption (IDE) approval for its Selution SLR to initiate its coronary pivotal clinical trial for in-stent restenosis. Geneva-based MedAlliance's Selution SLR is a drug-eluting balloon that is indicated for below-the-knee and superficial femoral artery (SFA) indications, for … [Read more...] about MedAlliance’s Selution SLR wins FDA IDE approval for in-stent restenosis
Catheters
Catheter Precision expands Vivo imaging system customer base in U.S.
Catheter Precision recently announced it expanded its U.S. customer base for the Vivo non invasive 3D imaging system. Vivo is a non-invasive 3D imaging system that allows physicians to identify the location of arrhythmias pre-procedure. It received FDA clearance at the end of 2020 and was strategically launched in the U.S. in 2021. Vivo has also … [Read more...] about Catheter Precision expands Vivo imaging system customer base in U.S.
NeoChord announces positive study results from transcatheter mitral chordal repair device
NeoChord recently announced positive results from a clinical study of its transcatheter NeXuS system for mitral chordal repair. The company's global early feasibility study treated patients with the transcatheter NeXuS system and results at one-year follow-up demonstrated notable clinical benefit and patient physiological improvement, according … [Read more...] about NeoChord announces positive study results from transcatheter mitral chordal repair device
Acutus Medical submits AcQBlate force-sensing ablation catheter for FDA premarket approval
Acutus Medical today announced it submitted pivotal clinical data of its AcQBlate force-sensing ablation catheter to the FDA for premarket approval. The Carlsbad, California-based company submitted data from the AcQForce Flutter trial, a prospective, multicenter, non-randomized study approved under an FDA investigational device exemption … [Read more...] about Acutus Medical submits AcQBlate force-sensing ablation catheter for FDA premarket approval
FDA clears Impella low-profile sheath from Abiomed
Abiomed (Nasdaq:ABMD) announced today that it received FDA 510(k) clearance for its Impella low-profile sheath. Danvers, Massachusetts-based Abiomed designed its sheath to maintain the same inner diameter as the existing 14 French (Fr) sheath used for Impella CP placement. However, the new sheath reduces the outer diameter by nearly 2 Fr. The … [Read more...] about FDA clears Impella low-profile sheath from Abiomed
Biosense Webster launches HelioStar in Europe
Biosense Webster today launched its HelioStar balloon ablation catheter in Europe. Johnson & Johnson's subsidiary Biosense Webster launched the radiofrequency balloon ablation catheter for catheter-based cardiac electrophysiological mapping of the atria when used with a multi-channel RF generator. According to the company, more than 11 … [Read more...] about Biosense Webster launches HelioStar in Europe
Ancora Heart appoints David Tucker as CFO
Ancora Heart today announced it appointed David Tucker as chief financial officer. Tucker will be responsible for the Santa Clara, California-based company's financial strategies and operations as it enrolls patients in the Corcinch-HF pivotal clinical trial of its AccuCinch ventricular restoration system. “David’s experience includes … [Read more...] about Ancora Heart appoints David Tucker as CFO
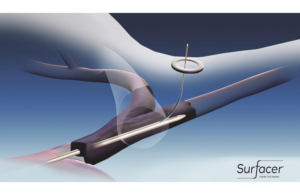
How this inside-out vascular access system simplifies catheter placement
Bluegrass Vascular Technologies is making catheter placement procedures for dialysis and chemotherapy easier and safer using inside-out technology, explains CEO and President Gabi Niederauer. Bluegrass Vascular Technologies has figured out a way to make a catheter sheath go inside the body and back out for easy catheter placement in standard … [Read more...] about How this inside-out vascular access system simplifies catheter placement
B. Braun buys catheter securement device portfolio from Starboard
B. Braun Medical announced today that it acquired the Clik-FIX catheter securement device portfolio from Starboard Medical. Clik-FIX adds to B.Braun’s offerings that help improve patient experiences and outcomes for peripheral intravenous (IV) therapy. Get the full story at our sister site, MassDevice. … [Read more...] about B. Braun buys catheter securement device portfolio from Starboard
Teleflex launches new injectable midline catheter portfolio
Teleflex (NYSE:TFX) announced today that it released its new Arrow pressure injectable midline portfolio of catheters in North America. Wayne, Pennsylvania-based Teleflex designed its Arrow portfolio to help alleviate line identification confusion. The company said a lack of identification between midlines and catheters creates the potential for … [Read more...] about Teleflex launches new injectable midline catheter portfolio