Abbott Vascular: Dilation catheters
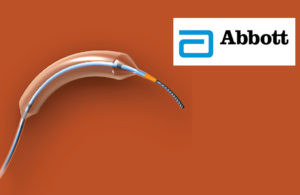
The recall involved 13,891 devices, which are used to open clogged blood vessels to improve blood flow to the heart. The devices were recalled because the balloons posed the risk of not deflating as intended due to weaker material close to the balloon bond resulting from excessive exposure to heat during manufacturing.
Use of the devices could have caused serious adverse health consequences, such as prolonged cardiac ischemia, air embolism, thrombosis, myocardial infarction and additional surgery that could lead to post-operative complications, including death.
Abbott Vascular received 13 complaints related to the devices with one reported death, according to a Feb. 19 FDA letter.
The company told healthcare providers to immediately stop using affected devices and return all unused affected products to Abbott Vascular.
Devices involved in the recall included models with balloon diameters of 4 mm, 4.5 mm and 5 mm. They were distributed between August 2019 and January 2020.

