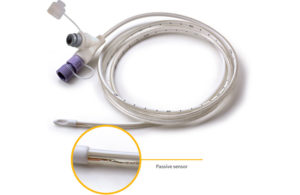
ENvizion Medical announced it has won the Frost & Sullivan 2021 North American Technology Innovation Leadership Award for its ENvue system. Israel-based ENvizion Medical has developed the feeding tube navigation system for accurate enteral tube placement in adult patients as an alternative to the limitations of blind insertions and the … [Read more...] about Envizion Medical wins Frost & Sullivan award
Resonetics acquires Distal Solutions
Resonetics today said it completed its acquisition of Distal Solutions for an undisclosed amount. Distal Solutions specializes in the design and development of catheter-based delivery systems for medical devices and implants. Distal's founder David Rezac will lead Resonetics's design and development efforts and California market … [Read more...] about Resonetics acquires Distal Solutions
Cardiovascular Systems launches peripheral balloon catheter
Cardiovascular Systems (NSDQ:CSII) today said it has launched its OrbusNeich Jade percutaneous transluminal angioplasty over-the-wire balloon catheter in the U.S. Jade is an over-the-wire (OTW) balloon catheter for the peripheral vasculature, including obstructed native arteries and synthetic arteriovenous dialysis fistulae and post-dilation of … [Read more...] about Cardiovascular Systems launches peripheral balloon catheter
Micro Interventional Devices tricuspid annuloplasty system wins FDA breakthrough device designation
Micro Interventional Devices today said it received FDA breakthrough device designation for its MIA-T percutaneous tricuspid annuloplasty system. The MIA-T system treats moderate to severe tricuspid regurgitation through a 12F catheter-based system. Micro Interventional Devices studied the safety and performance of the device in a study … [Read more...] about Micro Interventional Devices tricuspid annuloplasty system wins FDA breakthrough device designation
HeMo Bioengineering aspiration catheter wins pre-market approval in China
HeMo Bioengineering this week announced that it received pre-market approval in China for its aspiration catheter. The Afentta intracranial thrombectomy aspiration catheter is a direct aspiration treatment option for acute ischemic stroke thrombectomy and has demonstrated lower post-stroke treatment disability rates and disease burden in … [Read more...] about HeMo Bioengineering aspiration catheter wins pre-market approval in China
Pentax Medical Europe wins CE mark approval for single-use bronchoscope
Pentax Medical Europe today said it received CE mark approval for its single-use bronchoscope and is set to launch the device in Europe later this year. The Tokyo-based company designed the PulmoOne single-use bronchoscope to mimic the ergonomics of a reusable bronchoscope with similar scope handling and tip angulation that requires no change in … [Read more...] about Pentax Medical Europe wins CE mark approval for single-use bronchoscope
Prytime Medical gains approvals for catheter
Prytime Medical Devices today said it received U.K. and CE mark approval for its ER-REBOA Plus catheter. The Boerne, Texas-based company designed the ER-REBOA (resuscitative endovascular balloon occlusion of the aorta) catheter to improve the ease of use for controlling non-compressible truncal hemorrhage. "With this milestone, the new and … [Read more...] about Prytime Medical gains approvals for catheter
Boston Scientific gets CE mark approval for single-use bronchoscope
Boston Scientific today said it received CE mark approval for its Exalt Model B single-use bronchoscope. The Marlborough, Mass.–based company designed the device for bedside procedures in the intensive care unit, operating room and bronchoscopy suite. Exalt Model B is available in three sizes — slim, regular and large — for procedures such as … [Read more...] about Boston Scientific gets CE mark approval for single-use bronchoscope
FDA clears EndoFresh disposable digestive endoscope
EndoFresh today said it received FDA 510(k) clearance for its disposable digestive endoscopy system. The system features a camera system with an all-in-one design, disposable upper GI endoscope and disposable colonoscope, which can be used with the company's medical display and other peripheral devices for physicians to visualize, diagnose and … [Read more...] about FDA clears EndoFresh disposable digestive endoscope
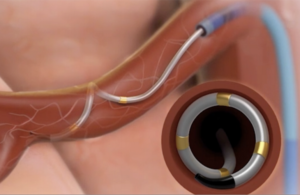
Is renal denervation back as a high blood pressure treatment?
Medtronic (NYSE:MDT) today reported clinically significant and sustained blood pressure reduction among nearly 3,000 people with uncontrolled hypertension treated with the company’s Symplicity renal denervation system. The study results, reported today out of the 2021 EuroPCR Annual Meeting, come the same week as ReCor Medical touted its … [Read more...] about Is renal denervation back as a high blood pressure treatment?