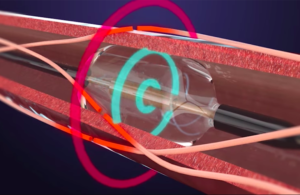
ReCor Medical‘s renal denervation significantly reduced blood pressure among people with drug-resistant hypertension, according to a new study reported at the American College of Cardiology’s 70th Annual Scientific Session. The study news, announced yesterday, could suggest a turnaround in fortunes for renal denervation technology. One … [Read more...] about ReCor Medical has positive renal denervation study news
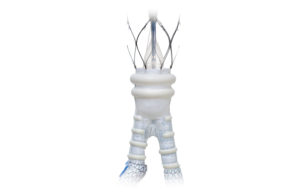
Edwards Lifesciences tricuspid valve reduced leakage in 98% of patients
Edwards Lifesciences‘s Evoque tricuspid valve replacement system reduces tricuspid heart valve leakage to non-traceable or mild leakage in 98% of patients, according to a new study reported at the American College of Cardiology’s 70th Annual Scientific Session. Evoque is designed to replace native tricuspid valves without open-heart surgery. The … [Read more...] about Edwards Lifesciences tricuspid valve reduced leakage in 98% of patients
Galaxy Medical, Japan Lifeline ink distribution agreement for ablation catheters
Galaxy Medical and Japan Lifeline today said they signed an exclusive distribution agreement for the Alpha1 ablation catheter. Under the agreement Galaxy will pursue U.S. regulatory approval for Japan Lifeline's Alpha1 in support of the commercialization of Galaxy's Centauri pulsed electric field system, which can be used with the ablation … [Read more...] about Galaxy Medical, Japan Lifeline ink distribution agreement for ablation catheters
BD enrolls first patients in arteriovenous endovascular fistula post-market trial
BD (NYSE: BDX) recently announced that it has begun enrollment for its post-market surveillance study of its WavelinQ arteriovenous endovascular fistula device. The Connect-AV trial is a prospective, single-arm, open-label study that will follow WavelinQ EndoAVF system patients for 24 months. Its dual primary effectiveness endpoints are the … [Read more...] about BD enrolls first patients in arteriovenous endovascular fistula post-market trial
Boston Scientific initiates coronary drug-coated balloon study in U.S.
Boston Scientific (NYSE:BSX) this week launched its Agent IDE trial for its Agent drug-coated balloon. The U.S. prospective, randomized clinical trial will evaluate the safety and effectiveness of a drug-coated balloon (DCB) in patients with coronary in-stent restenosis in lesions up to 26 mm in length in a coronary artery 2.0 mm to 4.0 mm in … [Read more...] about Boston Scientific initiates coronary drug-coated balloon study in U.S.
Endologix launches Alto abdominal stent graft in Canada
Endologix (NSDQ:ELGX) today said it launched its Alto abdominal stent graft in Canada following its recent Health Canada approval. The Alto abdominal stent graft system is a polymer-based therapy for abdominal aortic aneurysm (AAA) patients. It uses a low-profile delivery system with a conformable sealing ring that molds in-situ to the patient's … [Read more...] about Endologix launches Alto abdominal stent graft in Canada
NXgenPort, Mayo Clinic ink license agreement over chemo-port catheter for home monitoring
NXgenPort today announced that it has entered into a license agreement with Mayo Clinic to develop and test an implantable chemo-port catheter. NXgenPort's technology has chemo-port efficacy with sensor technology to measure and remotely monitor the early onset of complications by reporting and tracking patient response during … [Read more...] about NXgenPort, Mayo Clinic ink license agreement over chemo-port catheter for home monitoring
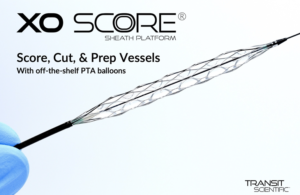
Transit Scientific scoring sheath wins CE mark
Transit Scientific today said it received CE mark clearance for its XO Score scoring sheath platform. XO Score is designed for dilation of stenotic material in the peripheral vasculature, including popliteal, infrapopliteal and native or synthetic arteriovenous dialysis fistulae. The scoring and cutting technology in the device enables … [Read more...] about Transit Scientific scoring sheath wins CE mark
iVascular covered stent gains CE mark
iVascular recently announced that it received CE mark approval for its BX-covered stent iCover. Balloon-expanding (BX) covered stents are used to treat arteriosclerotic lesions in renal and iliac arteries and to treat aneurysms and ruptures. The iCover stent adapts to tortuous vessels because of its high flexibility and post-expansion … [Read more...] about iVascular covered stent gains CE mark
FDA clears Olympus airway mobilescopes
Olympus (TYO:7733) today said it received FDA clearance for its line of airway mobilescopes. The mobilescopes enable physicians to perform a broad range of upper and lower airway management procedures. "We are proud to present to the market the only flexible mobilescope models with a video camera on the tip," Lynn Ray, VP and GM of global … [Read more...] about FDA clears Olympus airway mobilescopes