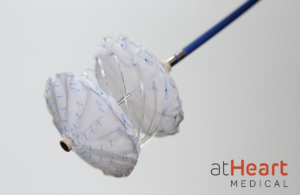
AtHeart Medical is initiating a U.S. investigational device exemption trial for its atrial septal defects (ASD) occluder. The reSept ASD Occluder is an occluder with a metal-free, bioresorbable frame. The implant is designed to overcome the limitations of occluders by reducing the risk of complications associated with the long-term presence of … [Read more...] about atHeart Medical launches atrial septal defect occluder trial
Seigla Medical gains CE mark for guide catheter extension
Startup Seigla Medical recently announced that it won CE mark approval for its LiquID guide catheter extension for use in coronary and peripheral vasculature interventions. Guide catheter extensions are used by cardiologists to assist in the delivery of stents and other interventional devices, according to the Minneapolis-based … [Read more...] about Seigla Medical gains CE mark for guide catheter extension
Contego Medical carotid stent wins CE mark approval
Contego Medical recently announced that it received CE mark approval for its Neuroguard IEP 3-in-1 carotid stent system and post-dilation balloon system with integrated embolic protection. The system has a nitinol stent, a pre-positioned post-dilation balloon and a microembolic filter with 40 μm pores. Its closed-cell stent is designed for … [Read more...] about Contego Medical carotid stent wins CE mark approval
Solo-Dex launches peripheral nerve block catheter
Solo-Dex this week launched its Fascile continuous peripheral nerve block catheter. The nerve block catheter's creators designed it to manage pain before, during and after surgery. It activates the facile continuous nerve block catheter quickly and safely for common procedures like shoulder repair, hip and knee replacement and other general … [Read more...] about Solo-Dex launches peripheral nerve block catheter
BD lands FDA clearance for hemodialysis catheter
BD (NYSE:BDX) this week said it has received FDA 510(k) clearance for its Pristine long-term hemodialysis catheter. The catheter has a side-hole free symmetric Y-tip distal lumen design that is designed to minimize thrombus adhesion and help facilitate blood clot aspiration prior to hemodialysis treatment. Its symmetric tip is designed to help … [Read more...] about BD lands FDA clearance for hemodialysis catheter
Endologix acquires PQ Bypass
Endologix today announced that it has completed its acquisition of PQ Bypass for an undisclosed amount. PQ Bypass makes the Detour platform that is designed for percutaneous femoral-popliteal bypass, which has been designated as a breakthrough device by the FDA. The Detour system has the company’s Torus stent graft and the PQ crossing device. … [Read more...] about Endologix acquires PQ Bypass
Qosina adds single-use bioprocessing devices
Qosina this week launched its product line for the single-use bioprocess industry. The new product line includes tube-to-tube bard connectors, luer fittings, check valves, tubing pinch clamps, tubing, steam thru connectors and more. “Qosina has been a trusted supplier for over 40 years and has worked with some of the best-known brands,” … [Read more...] about Qosina adds single-use bioprocessing devices
IceCure Medical wins FDA breakthrough device designation for cryoablation system
IceCure Medical today said it received FDA breakthrough device designation for its ProSense cryoablation system. ProSense is a liquid nitrogen-based cryoablation system that is a minimally invasive treatment for cancer tumors. It is designed for use in the treatment of TI invasive breast cancer or in patients not suitable for surgical … [Read more...] about IceCure Medical wins FDA breakthrough device designation for cryoablation system
4 innovative devices delivered through catheters
Physicians use catheters to deploy numerous life-saving devices across the healthcare realm, from cardiovascular to neurovascular. They typically insert the thin, flexible tubes in a process called catheterization to deploy devices such as left ventricular assist devices (LVADs), transcatheter aortic valve replacements (TAVRs) and occluders to … [Read more...] about 4 innovative devices delivered through catheters
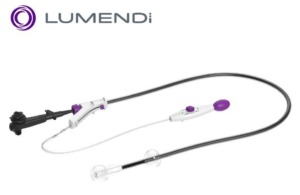
Lumendi launches hydrophilic coating for endoscopic devices
Lumendi today announced that it launched its EZ Glide hydrophilic coating for double-balloon endoluminal devices. The hydrophilic coating is designed for the inner sheath of the company's DiLumen EIP double-balloon endoluminal, which is used to improve the navigation of the endoscope through the bowel to create a stabilizing therapeutic zone … [Read more...] about Lumendi launches hydrophilic coating for endoscopic devices