Teleflex (NYSE:TFX) this week initiated a voluntary field safety corrective action in Europe for its Arrow subsidiary's multiple-lumen catheter. In a field safety notice dated March 16, Teleflex announced that Arrow is voluntarily recalling the catheter due to a potential sterility issue. The catheter's product tray may be damaged in affected … [Read more...] about Teleflex warns on Arrow multi-lumen catheter
Catheters
Reflow Medical lands 510(k) for expanded indication for Wingman catheter
Reflow Medical recently announced that it received FDA clearance for expanded indication of its Wingman Crossing Catheter. The Wingman Catheter is designed to cross peripheral CTOs using an extendable beveled tip that creates a channel to help penetrate or cross the occlusion was a guidewire. Reflow Medical received expanded indication for the … [Read more...] about Reflow Medical lands 510(k) for expanded indication for Wingman catheter
Abbott wins CE Mark for FlexNav delivery system
Abbott (NYSE:ABT) announced today that it won CE Mark approval in Europe for the FlexNav delivery system for its Portico transcatheter aortic valve implantation (TAVI) system. The Abbott Park, Ill.-based company’s FlexNav delivery system is designed to offer another tool for doctors to treat patients requiring a transcatheter aortic valve … [Read more...] about Abbott wins CE Mark for FlexNav delivery system
Zeus launches flexible extruded catheter liners
Zeus this week added a flexible extruded PTFE catheter liner to its StreamLiner product portfolio. With the addition, the company can extrude liners over-the-wire (OTW) in sizes comparable to film cast tubing. The StreamLiner OTW UT offers a normal wall thickness as low as 0.0004 in. with a tolerance of +/- 0.0002 in. "StreamLiner OTW bridges … [Read more...] about Zeus launches flexible extruded catheter liners
Access Vascular wins FDA clearance for HydroPICC catheter
Access Vascular this week received FDA clearance for its second-generation HydroPICC peripherally inserted central catheter. The HydroPICC has demonstrated thrombus accumulation rate 30 times less than traditional peripherally inserted central catheters (PICC), according to the company. PICCs can help reduce the risk of catheter occlusions … [Read more...] about Access Vascular wins FDA clearance for HydroPICC catheter
XableCath wins CE Mark approval for crossing catheters
XableCath recently announced its Crossing Catheters received CE Mark approval for peripheral use. The crossing devices come in blunt tip and abrasion tip versions with a variety of diameters and lengths. The company won FDA clearance for the devices in 2019. "I am pleased XableCath intravascular products continue to demonstrate a high degree … [Read more...] about XableCath wins CE Mark approval for crossing catheters
Levity Products launches external catheter stabilizers
Levity Products this week launched a set of newly patented external catheter stabilizers. The Levi External Catheter Stabilizer (LECS) device allows for a unique method of securing urine and balloon ports. The devices come in three variations designed for different types of catheters to improve clinical outcomes and quality of care. "Levity's … [Read more...] about Levity Products launches external catheter stabilizers
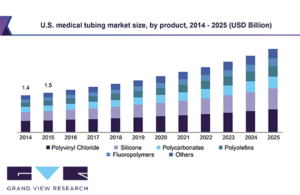
Medical tubing market to be worth $12 billion in 2025
The medical tubing market is expected to be worth $11.9 billion by 2025, according to an analysis from Grand View Research. Grand View Research suggests the industry will expand at a revenue-based consumer annual growth rate of 9.2% over the next five years. Increased health expenditures from individuals and increased emphasis on healthcare by … [Read more...] about Medical tubing market to be worth $12 billion in 2025
Ablative Solutions touts study of its infusion catheter for renal denervation
Ablative Solutions announced today that it received positive results at six months from its post-market study for the Peregrine System infusion catheter, which accomplishes renal denervation by delivering microdoses of dehydrated alcohol. The results from the study of the safety and efficacy of the CE-Marked Peregrine System have been published … [Read more...] about Ablative Solutions touts study of its infusion catheter for renal denervation
Bluegrass Vascular Technologies wins de novo nod for ‘inside-out’ vascular access catheter
Bluegrass Vascular Technologies announced that it won FDA de novo clearance for its Surfacer inside-out access catheter system. The Surfacer is designed to obtain central venous access through its inside-out approach, facilitating catheter insertion to the central venous system in people with upper body venous occlusions or other conditions that … [Read more...] about Bluegrass Vascular Technologies wins de novo nod for ‘inside-out’ vascular access catheter