MedAlliance announced today that Cordis acquired it for a total consideration that could reach up to $1.135 billion. The companies initially announced the planned deal in October of last year. Cordis, which develops interventional cardiovascular and endovascular technologies, made a $35 million investment last year. Its upfront closing … [Read more...] about Cordis acquires drug-eluting balloon maker MedAlliance for up to $1.135B
Catheters
Catheter supplier Zeus seeks $4B buyer, Reuters reports
Zeus is exploring the potential sale of its business in a deal that could be worth $4 billion, Reuters reported, citing "people familiar with the matter." The Orangeburg, South Carolina-based manufacturer of catheter components and other medtech supplies is reportedly working with Goldman Sachs to evaluate the upside of a deal with strategic … [Read more...] about Catheter supplier Zeus seeks $4B buyer, Reuters reports
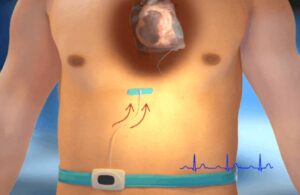
PercAssist treats first patient in heart failure support trial
PercAssist announced today that it successfully completed the first patient treatment in its EUREKA first-in-human clinical study. The study evaluates PercAssist's minimally invasive extravascular platform that provides hemodynamic support for chronic heart failure patients. Principal investigator Dr. Petr Neuzil completed the first case at … [Read more...] about PercAssist treats first patient in heart failure support trial
Boston Scientific’s Dr. Michael Jaff on the future of peripheral intervention
Advanced technologies have the potential to take peripheral therapy "to a different stratosphere," says Dr. Michael Jaff, the chief medical officer and VP of the Boston Scientific Peripheral Interventions business. He characterized them as similar to the ability to combine devices with drugs, which he called the biggest transformational change … [Read more...] about Boston Scientific’s Dr. Michael Jaff on the future of peripheral intervention
How LimFlow’s foot-saving system prevents amputations in patients with no other options
LimFlow’s breakthrough system for treating chronic limb-threatening ischemia (CLTI) is the first of its kind approved by the FDA for this severe form of peripheral artery disease (PAD). For CLTI patients who have lost blood flow below their knee and have no other suitable endovascular or surgical treatment options available, the LimFlow System … [Read more...] about How LimFlow’s foot-saving system prevents amputations in patients with no other options
Zeus opens Minnesota catheter manufacturing facility
Zeus has opened its catheter manufacturing facility in Arden Hills, Minnesota, celebrating the new facility with a grand opening. The 75,600-ft² building is home to CathX Medical, which Zeus acquired 2021. The facility has advanced R&D lab and cleanroom space for designing, developing and validating medical catheter prototypes. The … [Read more...] about Zeus opens Minnesota catheter manufacturing facility
Biosense Webster begins Omnypulse pulsed-field ablation trial
Johnson & Johnson's Biosense Webster today announced the first cases with its investigational Omnypulse catheter as part of the Omny-IRE clinical trial. The Omnypulse platform features the Omnypulse catheter and the Trupulse generator. Omny-IRE looks at the platform for mapping and treatment of symptomatic paroxysmal AFib during standard … [Read more...] about Biosense Webster begins Omnypulse pulsed-field ablation trial
CereVasc wins FDA IDE to expand eShunt trial
CereVasc announced today that the FDA approved an investigational device exemption (IDE) supplement for a study of its eShunt system. The FDA permitted the expansion of the study for patients with normal pressure hydrocephalus (NPH) to additional participants and sites. The company initiated the pilot study of its eShunt system under IDE in … [Read more...] about CereVasc wins FDA IDE to expand eShunt trial
Farapulse founder’s Field Medical raises $14M for next-gen cardiac ablation tech
Field Medical announced today that it closed an oversubscribed seed round with investments reaching $14 million. Private investors led the funding for the Cardiff-by-the-Sea, California-based cardiac ablation technology developer. Multiple strategic investors also joined the round. The company earmarked the funds to support … [Read more...] about Farapulse founder’s Field Medical raises $14M for next-gen cardiac ablation tech
FDA approves LimFlow chronic limb-threatening ischemia treatment
LimFlow announced today that the FDA approved its LimFlow System for treating chronic limb-threatening ischemia (CLTI). Approval enables the treatment to help those with CLTI with no other suitable endovascular or surgical treatment options available. These patients can face major amputation as a result of their condition. France-based … [Read more...] about FDA approves LimFlow chronic limb-threatening ischemia treatment