Olympus is warning health providers about incompatible laser use with bronchoscopes after receiving reports of endobronchial combustion, serious patient injury, and one death. The Japanese medtech giant said on July 3 that its voluntary field corrective action involves 32 BF series endoscope models, 19 of which it distributes in the U.S. The … [Read more...] about Olympus corrective action covers laser-compatible bronchoscopes
Recalls
Teleflex recall of Arrow catheter system is Class I
The FDA today designated Teleflex's recall of its Arrow Endurance extended dwell peripheral catheter system as Class I, the most serious type of medical device recall. The recall involved 262,016 devices distributed from October 26, 2018, to May 10, 2023, according to the FDA. Teleflex and its Arrow International subsidiary have reported 83 … [Read more...] about Teleflex recall of Arrow catheter system is Class I
Fresenius Kabi recall of Ivenix infusion systems in Class I
The FDA this week said the Fresenius Kabi recall of some Ivenix infusion systems is Class I, the most serious kind. Fresenius Kabi is recalling some Ivenix infusion systems due to a leak in the system that allows fluid to enter the administration set loading area near the air detector. The leak could damage the electrical system and result in … [Read more...] about Fresenius Kabi recall of Ivenix infusion systems in Class I
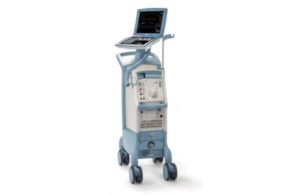
Getinge’s Datascope has its second Class I recall in a month
The FDA issued a notice determining another recall of Getinge subsidiary Datascope’s Cardiosave intra-aortic balloon pumps (IABPs) as Class I, the most serious kind. This recall relates to the Swedish medtech company’s Cardiosave Hybrid and Cardiosave Rescue IABPs. Both devices were subject to a separate Class I recall earlier this month. The … [Read more...] about Getinge’s Datascope has its second Class I recall in a month
Getinge’s Datascope has another serious intra-aortic balloon pump recall
The FDA issued a notice determining a recall of Getinge subsidiary Datascope's Cardiosave intra-aortic balloon pumps (IABPs) as Class I, the most serious kind. This recall relates to the Swedish medtech company's Cardiosave Hybrid and Cardiosave Rescue IABPs. Both devices were subject to a separate recall that the FDA determined was Class I in … [Read more...] about Getinge’s Datascope has another serious intra-aortic balloon pump recall
FDA sends warning letter to Olympus over endoscope manufacturing
The FDA issued Olympus a warning letter regarding adultered devices following an inspection of the company’s Tokyo facility. This marks the latest warning letter sent by the FDA to Olympus. The company received two separate letters at the end of last year. According to the letter, the agency conducted an inspection of the Tokyo facility … [Read more...] about FDA sends warning letter to Olympus over endoscope manufacturing
Getinge penalized in EU over life support system problems
The EU notified body DEKRA out of Germany has temporarily suspended the CE certificates for Getinge HLS and PLS sets, starting this month. The Swedish medtech company announced the suspension on Feb. 23. Health providers use the HLS and PLS for extracorporeal respiratory and/or cardiovascular support. Getinge had already announced potential … [Read more...] about Getinge penalized in EU over life support system problems
FDA labels Medtronic recall of some Mahurkar hemodialysis catheters as Class I
The FDA this week said the Medtronic (NYSE:MDT) recall of some Mahurkar hemodialysis catheters is Class I, the most serious kind. Medtronic is recalling its Mahurkar acute dual lumen high flow 13.5 Fr hemodialysis catheters, also known as Mahurkar QPlus, due to reports of a potential catheter hub defect that could cause leaks in a catheter's … [Read more...] about FDA labels Medtronic recall of some Mahurkar hemodialysis catheters as Class I
BD issues urgent field safety notice for Venovo venous stent system
BD has issued an urgent field safety notice for its Venovo venous stent system. The notice out of Germany warns of potential delayed deployment and silicone embolization if the proximal end of the Venovo venous stent system does not immediately expand upon deployment and remains connected to the stent cushion on the delivery system. According … [Read more...] about BD issues urgent field safety notice for Venovo venous stent system
FDA labels Medtronic recall of some endotracheal tubes as Class I
The FDA this week said the Medtronic recall of some NIM Contact Reinforced EMG endotracheal tubes is Class I, the most serious kind. Medtronic is recalling its NIM Contact Reinforced EMG endotracheal tubes and NIM Standard Reinforced EMG endotracheal tubes due to reports of obstruction of the endotracheal tube while in use in patients. Tube … [Read more...] about FDA labels Medtronic recall of some endotracheal tubes as Class I