The FDA issued healthcare providers updated guidance for certain warning language with paclitaxel-coated devices that treat PAD. These peripheral arterial disease (PAD)-treating devices produced data that does not support an excess mortality risk. Specifically, the FDA's guidance eliminates the need for certain warning language in the device … [Read more...] about FDA updates guidance on paclitaxel-coated devices, determines no link to late mortality risk
Catheters
BioCardia seeks Japanese approval for catheter-based heart failure cell therapy
BioCardia (Nasdaq:BCDA) announced today that it completed the submission of its CardiAMP cell therapy system to Japan’s regulatory authorities. Sunnyvale, California-based BioCardia submitted CardiAMP to Japan’s Pharmaceutical and Medical Device Agency (PMDA). It seeks approval for the indication of ischemic heart failure with reduced ejection … [Read more...] about BioCardia seeks Japanese approval for catheter-based heart failure cell therapy
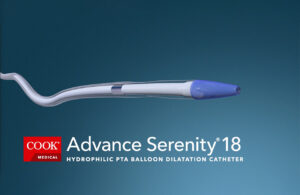
Cook Medical has more hydrophilic PTA balloon catheter options
Cook Medical today announced more sizes and locations for the Advance Serenity hydrophilic PTA balloon catheter product line. It's now possible for U.S. and Canadian interventionalists to use Advance Serenity for below-the-knee and above-the-knee procedures to treat patients with peripheral artery disease (PAD), according to Cook Medical. In … [Read more...] about Cook Medical has more hydrophilic PTA balloon catheter options
Teleflex recall of Arrow catheter system is Class I
The FDA today designated Teleflex's recall of its Arrow Endurance extended dwell peripheral catheter system as Class I, the most serious type of medical device recall. The recall involved 262,016 devices distributed from October 26, 2018, to May 10, 2023, according to the FDA. Teleflex and its Arrow International subsidiary have reported 83 … [Read more...] about Teleflex recall of Arrow catheter system is Class I
FDA approves Metavention’s renal denervation for hypertension pivotal IDE study
Metavention announced today that the FDA granted approval to initiate an investigational device exemption (IDE) trial for its renal denervation system. Minneapolis-based Metavention designed its integrated radiofrequency (iRF) renal denervation system to treat hypertension. The company plans for its randomized, double-blinded, sham-controlled … [Read more...] about FDA approves Metavention’s renal denervation for hypertension pivotal IDE study
Freudenberg Medical implements VR in training, reduces scrap materials
Freudenberg Medical recently announced it implemented virtual reality training for employees working in catheter production at operations in Ireland and the U.S. The VR training program will help ensure employees have the necessary skills to meet the high-quality standards in catheter manufacturing for customers worldwide, according to the … [Read more...] about Freudenberg Medical implements VR in training, reduces scrap materials
FDA clears Pounce LP thrombectomy system from Surmodics
Surmodics (Nasdaq:SRDX) today announced that it received FDA 510(k) clearance for its Pounce low-profile (LP) thrombectomy system. News of the regulatory nod comes just one day after Needham analysts upgraded Surmodics stock from "Hold" to "Buy." The analysts cited positive developments pointing toward the likely approval of Surmodics' SurVeil … [Read more...] about FDA clears Pounce LP thrombectomy system from Surmodics
Confluent Medical now offers EU MDR-compliant polyimide tubing
Confluent Medical Technologies announced today that it now offers REACH and EU MDR-compliant polyimide tubing. REACH, a European Union regulation, imposes stringent limitations for the EU market. It covers the amount of restricted substances contained in devices sold in the EU. It stands for Registration, Evaluation, Authorization, and … [Read more...] about Confluent Medical now offers EU MDR-compliant polyimide tubing
Merit Medical spends $132.5M on acquisitions to expand its offerings
AngioDynamics (Nasdaq:ANGO) announced that it completed the sale of its dialysis product portfolio to Merit Medical Systems. It was part of a series of product acquisitions worth $132.5 million that Merit (Nasdaq: MMSI) has announced. Latham, New York-based AngioDynamics sold its dialysis portfolio and the BioSentry product for $100 million … [Read more...] about Merit Medical spends $132.5M on acquisitions to expand its offerings
How Medtronic uses nitinol to improve the structure and effectiveness of heart devices
Tim Laske, VP of research and business development for Medtronic‘s cardiac ablation solutions business, discusses the challenges of designing devices for the heart and explores the properties of nitinol. The heart is often one of the most underappreciated aspects of human anatomy, and its atrial appendages are often overlooked even in cardiac … [Read more...] about How Medtronic uses nitinol to improve the structure and effectiveness of heart devices