Medtronic (NYSE:MDT) announced today that it voluntarily recalled a subset of its In.Pact Admiral and In.Pact AV balloon catheters. The company initiated the recent recall of the paclitaxel-coated percutaneous transluminal angioplasty (PTA) balloon catheters due to the potential for pouch damage resulting in a loss of sterility. Get the full … [Read more...] about Medtronic issues voluntary recall for certain In.Pact drug-coated balloon catheters
FDA approves Edwards Lifesciences’ tissue valve replacement for heart’s mitral position
Edwards Lifesciences (NYSE:EW) announced today that the FDA approved its Mitris Resilia tissue valve replacement system. Irvine, California-based Edwards designed the Mitris Resilia valve with a saddle-shaped sewing cuff that mimics the asymmetric shape of the native mitral valve. It features a low-profile frame for avoiding obstruction of the … [Read more...] about FDA approves Edwards Lifesciences’ tissue valve replacement for heart’s mitral position
Confluent Medical Technologies closes majority investment from TPG Capital
Confluent Medical Technologies announced that it closed a majority investment from TPG Capital following TPG’s agreement to acquire a majority stake in the company. Last month, Confluent Medical confirmed that TPG Capital signed a definitive agreement to acquire a majority stake in the company, joining existing investor Ampersand Capital … [Read more...] about Confluent Medical Technologies closes majority investment from TPG Capital
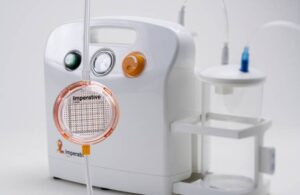
Imperative Care launches Zoom POD for stroke treatment
Imperative Care announced today that it launched its Zoom POD aspiration tubing sterile field clot filter device. Campbell, California-based Imperative Care designed the Zoom POD to enable faster time to clot capture during critical mechanical thrombectomy procedures for ischemic stroke patients. The company touts Zoom POD as the first and only … [Read more...] about Imperative Care launches Zoom POD for stroke treatment
Medtronic’s recall of HawkOne system is Class I
The FDA today issued a notice confirming that the recall of Medtronic’s (NYSE:MDT) HawkOne system is Class I, the most serious kind. So far in 2022, the FDA has classified three Medtronic-related recalls as Class I, with the Covidien (part of Medtronic) Puritan Bennett 980 series ventilator and the Synergy Cranial and StealthStation S7 Cranial … [Read more...] about Medtronic’s recall of HawkOne system is Class I
Alcon closes Ivantis acquisition
Alcon (NYSE:ALC) announced today that it completed its $475 million acquisition of glaucoma surgery device maker Ivantis. Geneva, Switzerland-based Alcon initially announced the acquisition of Ivantis, which develops the Hydrus Microstent minimally invasive glaucoma surgery (MIGS) device, in November 2021. According to a news release, the … [Read more...] about Alcon closes Ivantis acquisition
Medtronic to acquire cardiac arrhythmia treatment developer Affera
Medtronic (NYSE:MDT) announced today that it agreed to acquire cardiac mapping and ablation technology developer Affera. Newton, Massachusetts-based Affera designs and manufactures cardiac mapping and navigation systems, as well as catheter-based cardiac ablation technologies, including a differentiated, focal pulsed-field ablation platform for … [Read more...] about Medtronic to acquire cardiac arrhythmia treatment developer Affera
VitalPath acquires Modern Catheter Technologies
VitalPath announced today that it acquired catheter delivery system developer Modern Catheter Technologies. The Roseville, Minnesota-based company said in a news release that the acquisition builds upon its acquisition of VitalDyne in March 2021. Get the full story at our sister site, Medical Design & Outsourcing. … [Read more...] about VitalPath acquires Modern Catheter Technologies
Laborie acquires exclusive license for drug-coated baloon treatment for urethral strictures
Laborie Medical Technologies announced today that it acquired an exclusive license to the Optilume urethral drug-coated balloon (DCB). Portsmouth, New Hampshire-based Laborie purchased a perpetual, exclusive license to Optilume — developed by Urotronic — following FDA approval in December 2021. The DCB platform received CE mark approval for use … [Read more...] about Laborie acquires exclusive license for drug-coated baloon treatment for urethral strictures
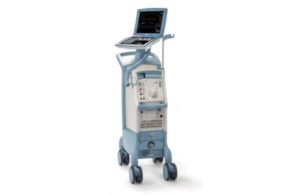
Getinge intra-aortic balloon pump recall is Class I
The FDA determined that Getinge's recall of its Cardiosave Hybrid and CardioSave Rescue devices is Class I, the most serious kind. Datascope/Getinge/Maquet designed the CardioSave Hybrid and CardioSave Rescue intra-aortic balloon pumps (IABP) as cardiac assist devices for patients undergoing cardiac and non-cardiac surgery. They are used in … [Read more...] about Getinge intra-aortic balloon pump recall is Class I