Ambu today said it received Health Canada clearance for its aScope 4 Cysto single-use cystoscope. The cystoscope is a flexible, single-use endoscope that is designed to eliminate significant capital, repair and cleaning costs with a patient-ready instrument for every procedure, according to Ambu. The device is used for diagnosing, managing and … [Read more...] about Ambu nabs Health Canada clearance for single-use cystoscope
Catheters
Red One Medical, Quick Tube Medical partner to deliver chest tubes to U.S. military
Red One Medical and Quick Tube Medical this week announced a partnership to provide chest tube devices to U.S. military members. Quick Tube Medical's chest tube system has a rapid deployment system for an average insertion to completion time of 30 to 45 seconds compared to eight minutes for some traditional chest tubes. "This cutting-edge … [Read more...] about Red One Medical, Quick Tube Medical partner to deliver chest tubes to U.S. military
Fresenius Kabi launches sodium chloride injection for maintaining catheter patency
Fresenius Kabi announced today that it launched two new presentations of Heparin sodium in its Freeflex IV bags. Lake Zurich, Ill.–based Fresenius Kabi’s Heparin sodium in sodium chloride injection at a concentration of two USP units per ml is indicated as an anticoagulant to maintain catheter patency, according to a news release. Get the full … [Read more...] about Fresenius Kabi launches sodium chloride injection for maintaining catheter patency
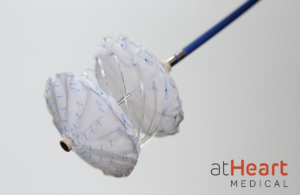
atHeart Medical launches atrial septal defect occluder trial
AtHeart Medical is initiating a U.S. investigational device exemption trial for its atrial septal defects (ASD) occluder. The reSept ASD Occluder is an occluder with a metal-free, bioresorbable frame. The implant is designed to overcome the limitations of occluders by reducing the risk of complications associated with the long-term presence of … [Read more...] about atHeart Medical launches atrial septal defect occluder trial
Seigla Medical gains CE mark for guide catheter extension
Startup Seigla Medical recently announced that it won CE mark approval for its LiquID guide catheter extension for use in coronary and peripheral vasculature interventions. Guide catheter extensions are used by cardiologists to assist in the delivery of stents and other interventional devices, according to the Minneapolis-based … [Read more...] about Seigla Medical gains CE mark for guide catheter extension
Contego Medical carotid stent wins CE mark approval
Contego Medical recently announced that it received CE mark approval for its Neuroguard IEP 3-in-1 carotid stent system and post-dilation balloon system with integrated embolic protection. The system has a nitinol stent, a pre-positioned post-dilation balloon and a microembolic filter with 40 μm pores. Its closed-cell stent is designed for … [Read more...] about Contego Medical carotid stent wins CE mark approval
FDA clears Acutus Medical suite of universal transseptal crossing devices
Acutus Medical announced that it received FDA clearance for its AcQCross family of universal transseptal crossing devices. Carlsbad, Calif.-based Acutus touts the AcQCross system as the first and only transseptal puncture system specifically engineered to pair and mate seamlessly with Acutus' suite of sheaths, as well as with sheaths sold by … [Read more...] about FDA clears Acutus Medical suite of universal transseptal crossing devices
Solo-Dex launches peripheral nerve block catheter
Solo-Dex this week launched its Fascile continuous peripheral nerve block catheter. The nerve block catheter's creators designed it to manage pain before, during and after surgery. It activates the facile continuous nerve block catheter quickly and safely for common procedures like shoulder repair, hip and knee replacement and other general … [Read more...] about Solo-Dex launches peripheral nerve block catheter
BD lands FDA clearance for hemodialysis catheter
BD (NYSE:BDX) this week said it has received FDA 510(k) clearance for its Pristine long-term hemodialysis catheter. The catheter has a side-hole free symmetric Y-tip distal lumen design that is designed to minimize thrombus adhesion and help facilitate blood clot aspiration prior to hemodialysis treatment. Its symmetric tip is designed to help … [Read more...] about BD lands FDA clearance for hemodialysis catheter
Endologix acquires PQ Bypass
Endologix today announced that it has completed its acquisition of PQ Bypass for an undisclosed amount. PQ Bypass makes the Detour platform that is designed for percutaneous femoral-popliteal bypass, which has been designated as a breakthrough device by the FDA. The Detour system has the company’s Torus stent graft and the PQ crossing device. … [Read more...] about Endologix acquires PQ Bypass