NXgenPort today announced that it has entered into a license agreement with Mayo Clinic to develop and test an implantable chemo-port catheter. NXgenPort's technology has chemo-port efficacy with sensor technology to measure and remotely monitor the early onset of complications by reporting and tracking patient response during … [Read more...] about NXgenPort, Mayo Clinic ink license agreement over chemo-port catheter for home monitoring
Catheters
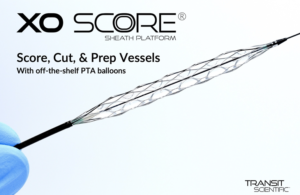
Transit Scientific scoring sheath wins CE mark
Transit Scientific today said it received CE mark clearance for its XO Score scoring sheath platform. XO Score is designed for dilation of stenotic material in the peripheral vasculature, including popliteal, infrapopliteal and native or synthetic arteriovenous dialysis fistulae. The scoring and cutting technology in the device enables … [Read more...] about Transit Scientific scoring sheath wins CE mark
Confluent Medical invests in Electrospinning Co.
Confluent Medical this week announced a strategic investment in the U.K.-based Electrospinning Co. Electrospinning Co. has developed an electrospinning process that will help Confluent Medical expand design options for the structural heart market and fully automate the process of attaching biomedical textiles to heart valve frames. “The … [Read more...] about Confluent Medical invests in Electrospinning Co.
Penumbra slides on Street-beating Q1 results
Penumbra (NYSE:PEN) yesterday posted first-quarter results that beat the earnings consensus on Wall Street and matched sales estimates. The Alameda, Calif.-based company reported profits of $11.836 million, or 32¢ per share, on sales of $169.2 for the three months ended March 31, for a bottom-line gain of 730.6% sales growth of 23.2% compared … [Read more...] about Penumbra slides on Street-beating Q1 results
Perfuze wins CE mark approval for clot aspiration catheter, treats first patients
Perfuze today said it received CE mark approval for its clot aspiration catheter Millipede 088. The Ireland-based company also announced that the first five acute ischemic stroke patients have been treated with the device. “Our initial experience with the device has been very positive, I believe the large bore and distal flow control … [Read more...] about Perfuze wins CE mark approval for clot aspiration catheter, treats first patients
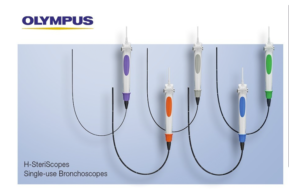
Olympus launches five single-use bronchoscopes
Olympus today said it has launched its product line of five H-SteriScope single-use bronchoscopes. The single-use endoscopes are for diagnostic and therapeutic procedures. They resulted from a collaboration between Olympus subsidiary Veran Medical Technologies and Hunan Vathin Medical Instrument. "We are very excited to launch our first line … [Read more...] about Olympus launches five single-use bronchoscopes
LeMaitre profits up 86% in Q1 results
LeMaitre Vascular (NSDQ:LMAT) posted first-quarter results yesterday that beat the consensus on Wall Street. The company reported profits of $5.9 million, or 28¢ per share, on sales of $35.8 million, for the three months ended March 31, for a bottom-line gain of 86.8% sales growth of 17.45% compared with Q1 2020. Get the full story on our … [Read more...] about LeMaitre profits up 86% in Q1 results
4 catheter-based devices to watch for in 2021
Medical device companies are working to gain regulatory approvals and launch new catheter-based devices as the world slowly returns to normal after reeling from the COVID-19 pandemic. As of April 19, the FDA had approved 11 medical devices this year — two of them catheter-based. In comparison, the regulatory agency approved 19 devices by April … [Read more...] about 4 catheter-based devices to watch for in 2021
Medtronic wins FDA breakthrough nod for Emprint ablation catheter kit
Medtronic (NYSE:MDT) announced today that it received FDA breakthrough device designation for its Emprint ablation catheter kit. Fridley, Minn.-based Medtronic's catheter is designed to be used in conjunction with the Emprint microwave generator and the Medtronic lung navigation platform to offer a minimally invasive, localized treatment of … [Read more...] about Medtronic wins FDA breakthrough nod for Emprint ablation catheter kit
FDA says Cordis carotid artery stent recall is serious
The FDA has classified Cordis‘ recall of its Precise Pro Rx carotid system as Class I, the most serious kind of recall. Cordis in February recalled certain lots of its Precise Pro RX carotid stent systems because of a risk of separation of the atraumatic distal tip of the sheathed delivery system in patients. Get the full story at our sister … [Read more...] about FDA says Cordis carotid artery stent recall is serious