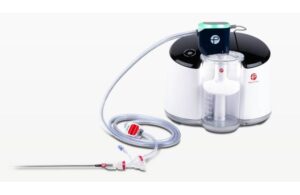
Gradient Denervation Technologies announced today that the FDA accepted it into its Total Product Life Cycle Advisory Program (TAP). TAP provides early and frequent strategic engagement from the FDA, patients, providers and payers. It facilitates rapid development and widespread access to medical devices. The company expects its acceptance into … [Read more...] about FDA adds Gradient denervation tech to advisory program
Catheters
Penumbra completes enrollment in pulmonary embolism trial
Penumbra (NYSE:PEN) announced today that it completed enrollment in the STORM-PE trial of its Lightning Flash system. STORM-PE evaluates Lightning Flash computer-assisted vacuum thrombectomy (CAVT) plus anticoagulation. It pits that against anticoagulation alone in the treatment of acute intermediate-high risk pulmonary embolism (PE). The study … [Read more...] about Penumbra completes enrollment in pulmonary embolism trial
Philips reports first cases for VeriSight Pro 3D ICE catheter in Europe
Philips today announced the first procedures in Europe with its recently launched VeriSight Pro 3D ICE catheter. Doctors at the German University Medical Center Mainz and Robert Bosch Hospital in Stuttgart completed the first procedures in Europe after the intracardiac echocardiography (ICE) catheter following its launch across the continent last … [Read more...] about Philips reports first cases for VeriSight Pro 3D ICE catheter in Europe
Teleflex has positive Arrow catheter data
Teleflex reports new findings from a study showing the efficacy of its Arrow chlorhexidine-impregnated central venous catheters (CVCs). The prospective study cohort included more than 6,670 patients from 12 intensive care units (ICUs). It spanned eight hospitals across India, Malaysia, Papua New Guinea, Colombia, Egypt and Turkey. Wayne, … [Read more...] about Teleflex has positive Arrow catheter data
Aortyx raises $16M for bioresorbable aortic patch
Aortyx announced today that it brought in $16 million (€13.8 million) to support its bioresorbable aortic patch technology. The company expects the funding to help bring the patch to first-in-human tests within two years. Funding came from Ship2B Ventures, through its BSocial Impact Fund (supported by Banco Sabadell, the EIF, and AXIS), and Clave … [Read more...] about Aortyx raises $16M for bioresorbable aortic patch
Atraverse raises $29.4M for left-heart access device
Atraverse Medical announced today that it closed a $29.4 million follow-on financing round to help accelerate growth. The San Diego-based company secured an oversubscribed seed funding round worth $12.5 million last year. This follow-on funding builds on that round, which helped to obtain FDA 510(k) clearance and begin the early rollout of … [Read more...] about Atraverse raises $29.4M for left-heart access device
FastWave Medical reports first-in-human use of coronary laser IVL system
FastWave Medical has completed the first human procedures with its Sola coronary laser intravascular lithotripsy system. The company said that these initial procedures marked the start of its multi-center feasibility study evaluating the platform’s safety and performance. [Scott Nelson, FastWave Medical’s co-founder and CEO, will participate in a … [Read more...] about FastWave Medical reports first-in-human use of coronary laser IVL system
Terumo Neuro launches Sofia Flow 88 for stroke care, expanding aspiration catheter line
Terumo Neuro (formerly MicroVention) has launched the Sofia Flow 88 large-bore aspiration catheter, designed for supporting clot removal in stroke procedures. The catheter features a 0.088 in. inner diameter and incorporates design enhancements aimed at improving procedural control, vessel safety, and compatibility across aspiration … [Read more...] about Terumo Neuro launches Sofia Flow 88 for stroke care, expanding aspiration catheter line
Reflow Medical wins FDA de novo clearance for Spur stent
Reflow Medical today announced it received FDA de novo clearance for its Spur Peripheral Retrievable Stent System for infrapopliteal arterial disease. The system is cleared for use in de novo or restenotic lesions following predilatation in below-the-knee (BTK) vessels. It combines a self-expanding stent with an integrated balloon catheter on an … [Read more...] about Reflow Medical wins FDA de novo clearance for Spur stent
Terumo begins limited release of Roadsaver carotid stent following FDA nod
Terumo Interventional Systems has announced the early commercial availability of its FDA-approved Roadsaver Carotid Stent System. Roadsaver is a dual-layer micromesh carotid stent designed to reduce embolic risk during carotid artery stenting (CAS) procedures. It is indicated for use in patients with carotid artery stenosis who are at increased … [Read more...] about Terumo begins limited release of Roadsaver carotid stent following FDA nod