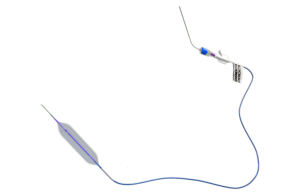
Cook Medical today commercially launched its Hercules 100 Transnasal Esophageal Balloon in the U.S. The balloon is designed specifically for trans nasal esophageal procedures and is another tool for ENT physicians to help treat esophageal strictures. "This balloon was specifically designed with ENT physicians and their patients in mind. The … [Read more...] about Cook Medical announces U.S. launch of balloon for transnasal esophageal procedures
Implants
VentureMed vessel prep system wins new FDA indication to treat in-stent restenosis
VentureMed recently announced it received FDA 510(k) clearance for its Flex Vessel Prep System for use in treating in-stent restenosis in the peripheral vasculature. The Flex Vessel Prep System modifies plaque and fibrous stenoses and creates controlled-depth micro-incisions of any length to release circumferential tension and improve vessel … [Read more...] about VentureMed vessel prep system wins new FDA indication to treat in-stent restenosis
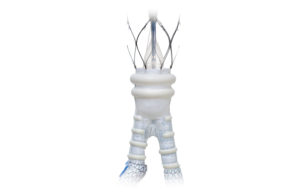
Endologix launches abdominal stent graft in Europe
Endologix today announced that it has launched its Alto abdominal stent graft system in Europe. The first implant of the stent graft took place in Stevenage, England, following the company's recent CE mark approval. The device was chosen for that specific patient because of their short, thrombus-lined conical aortic neck. “We are pleased to … [Read more...] about Endologix launches abdominal stent graft in Europe
Micro Medical gains CE Mark for stent and balloon
Micro Medical this week announced that it received CE Mark approval for its long over-the-wire balloon catheter and MicroStent vascular stent. MicroStent and MicroBalloon XL are part of the company's MMS Micro Vascular Integrated Platform and are designed to achieve and maintain vessel latency and improve blood flow to reduce below-the-knee … [Read more...] about Micro Medical gains CE Mark for stent and balloon
Medtronic Resolute Onyx stent gains expanded FDA clearance
Medtronic today said it received FDA clearance for an expanded indication of its Resolute Onyx drug-eluting stent (DES). The new indication allows for one-month of dual-antiplatelet therapy (DAPT) labeling with an expanded indication for high bleeding risk patients that are implanted with the device. Resolute Onyx is the only DES in the U.S. … [Read more...] about Medtronic Resolute Onyx stent gains expanded FDA clearance
Teleflex wins FDA clearance for EZ-IO intraosseous vascular access system expanded indications
Teleflex today said it received FDA 510(k) clearance for expanded indications of its Arrow EZ-IO intraosseous vascular access system. The device is designed to be used when intravenous access is difficult or impossible to obtain in emergent, urgent or medically necessary cases. The expanded indication allows the device to be used for up to 48 … [Read more...] about Teleflex wins FDA clearance for EZ-IO intraosseous vascular access system expanded indications
W. L. Gore debuts lower-profile endoprosthesis
W.L. Gore this week announced it has launched its lower profile, large diameter Gore ViaBahn Endoprosthesis. The device design reduces the delivery profile for larger 9-13 mm diameter stent grafts by up to 3 Fr to enable delivery through smaller sheaths. The smaller introducer sheaths result in a lower risk of vascular access complications and … [Read more...] about W. L. Gore debuts lower-profile endoprosthesis
Study: Medtronic’s drug-coated balloon reduces dialysis interruptions
Medtronic (NYSE:MDT) recently announced the results of a study showing that some kidney disease patients who had a drug-coated balloon (DCB) implanted to treat arteriovenous (AV) failure experienced fewer interruptions of dialysis treatment. Many end-stage renal disease (ESRD) patients require AV fistulae in order to receive continuous dialysis. … [Read more...] about Study: Medtronic’s drug-coated balloon reduces dialysis interruptions
What is medical tubing used for?
Medical tubing allows clinicians to administer fluid and or even devices — as well as potentially allow for gas flow. And that's just the beginning. Common applications of medical tubing include ventilators and IVs, but tubing also finds uses supporting access devices and as a delivery method for other devices. For example, tubing can be used to … [Read more...] about What is medical tubing used for?
Abbott wins CE Mark for FlexNav delivery system
Abbott (NYSE:ABT) announced today that it won CE Mark approval in Europe for the FlexNav delivery system for its Portico transcatheter aortic valve implantation (TAVI) system. The Abbott Park, Ill.-based company’s FlexNav delivery system is designed to offer another tool for doctors to treat patients requiring a transcatheter aortic valve … [Read more...] about Abbott wins CE Mark for FlexNav delivery system