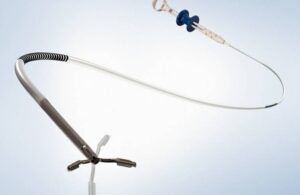
Second Heart Assist today announced data from a study highlighting the performance of its Whisper device against a competitive heart pump. The study showed that Whisper — a next-generation percutaneous mechanical circulatory support (pMCS) system — caused significantly less hemolysis than the widely used Johnson & Johnson MedTech/Abiomed … [Read more...] about Second Heart Assist says Whisper device caused less hemolysis compared to Abiomed Impella CP
Medtronic says new study backs Prevail paclitaxel-coated balloon
Medtronic (NYSE: MDT) announced results from a registry highlighting the effectiveness of the Prevail drug-coated balloon (DCB). Dr. Sacharias von Koch of the Deptartment of Cardiology and Clinical Sciences at Lund University, Skåne University Hospital in Sweden presented findings from the Swedish Coronary Angiography and Angioplasty Registry … [Read more...] about Medtronic says new study backs Prevail paclitaxel-coated balloon
Lungpacer wins FDA IDE for AeroNova system
Lungpacer Medical announced today that it received FDA investigational device exemption (IDE) to begin a trial for its AeroNova system. The STARI (stimulation to activate respiration) trial evaluates the feasibility of the investigational AeroNova system. It looks at the system in patients suffering from moderate to severe Acute Hypoxemic … [Read more...] about Lungpacer wins FDA IDE for AeroNova system
Reflow Medical expands global reach with European subsidiary
Reflow Medical announced today that it opened a European subsidiary in Landsberg am Lech, Germany, to expand outside the U.S. The company said its strategic expansion strengthens its international presence and enhances its ability to serve markets globally. SVP and GM Knut Saurteig leads the subsidiary, called Reflow Medical Europe GmbH. … [Read more...] about Reflow Medical expands global reach with European subsidiary
Stryker’s Inari Medical launches Artix thrombectomy system
Inari Medical — now part of Stryker — announced that it launched its Artix thrombectomy system for arterial thrombectomies. The company built Artix for the distinct needs of the peripheral arterial system, according to a news release. It marks Inari's inaugural entry into the arterial space. The combined aspiration plus mechanical … [Read more...] about Stryker’s Inari Medical launches Artix thrombectomy system
FTC challenges private equity firm’s acquisition of Surmodics
The U.S. Federal Trade Commission (FTC) has sued to block the planned acquisition of medical device company Surmodics by GTCR. According to a news release, GTCR also owns a majority stake in Biocoat. The FTC says Biocoat is the second-largest provider of outsourced hydrophilic medical device coatings, behind only Surmodics. As a result, the FTC … [Read more...] about FTC challenges private equity firm’s acquisition of Surmodics
Olympus launches single-use hemostasis clip
Olympus announced that it launched a new hemostasis clip to meet the needs of gastrointestinal (GI) endoscopists. The company's Retentia HemoClip offers 360° rotation and an intuitive one-step deployment. This provides control over placement with three different sizes to accommodate a variety of closure applications. Retentia HemoClip … [Read more...] about Olympus launches single-use hemostasis clip
J&J’s Shockwave Medical launches new IVL catheter
Johnson & Johnson MedTech‘s Shockwave Medical today announced the U.S. launch of its Javelin peripheral intravascular lithotripsy (IVL) catheter. The system is designed to modify calcium and cross extremely narrowed vessels in patients with peripheral artery disease (PAD). Shockwave said its novel, non-balloon IVL platform delivers a similar … [Read more...] about J&J’s Shockwave Medical launches new IVL catheter
Boston Scientific to acquire renal denervation company SoniVie
Boston Scientific (NYSE:BSX) announced today that it agreed to acquire intravascular ultrasound system developer SoniVie. Marlborough, Massachusetts-based Boston Scientific is a strategic investor in SoniVie, holding about a 10% equity stake. With this stake, the transaction features an upfront payment of approximately $360 million for the 90% … [Read more...] about Boston Scientific to acquire renal denervation company SoniVie
Olympus has a serious endoscope guide sheath recall
The FDA today issued a notice warning of the removal of certain single-use guide sheath kits for endoscope instruments by Olympus. This recall involves removing certain devices from where they are used or sold. The FDA identified it as the most serious type of recall, as it may cause serious injury or death. Olympus warned customers to not … [Read more...] about Olympus has a serious endoscope guide sheath recall