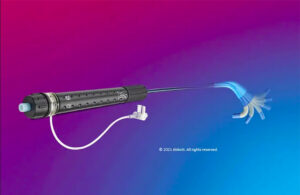
Acutus Medical (Nasdaq:AFIB) reported positive second-quarter results, with some future plans that got analysts excited. Shares of AFIB rose 0.8% at 68¢ apiece in mid-afternoon trading today. MassDevice's MedTech 100 Index — which includes stocks of the world's largest medical device companies — fell 2.1%. BTIG analysts Marie Thibault and Sam … [Read more...] about Acutus expects AcQBlate ablation catheter approval by end of 2023
Regulatory
Abbott has a serious recall involving its Amplatzer delivery sheath
The FDA today said an Abbott recall involving its Amplatzer steerable delivery sheath is Class I, its most serious level. Launched in the U.S. in April 2022, the Amplatzer sheath delivers Abbott's Amplatzer Amulet left atrial appendage occluder. Competing against Boston Scientific's Watchman, Amplatzer Amulet provides complete occlusion in order … [Read more...] about Abbott has a serious recall involving its Amplatzer delivery sheath
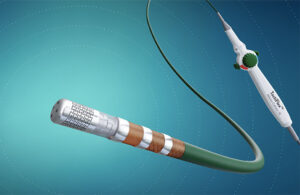
J&J’s Biosense Webster launches AFib mapping catheter
Johnson & Johnson's (NYSE: JNJ) Biosense Webster announced today that it launched the Optrell mapping catheter with TrueRef technology. Irvine, California-based Biosense Webster designed Optrell as a high-density diagnostic catheter powered by its Carto 3 system. It features small electrodes arranged in a fixed array formation. These provide … [Read more...] about J&J’s Biosense Webster launches AFib mapping catheter
After recall and relaunch, Medtronic wants to go global with its catheter-delivered Harmony valve
Each Medtronic Harmony valve is sewn by hand to attach laser-cut pig tissue to the nitinol that makes this minimally invasive heart implant possible. Medtronic’s Harmony transcatheter pulmonary valve (TPV) design is paying off after engineers solved a delivery catheter recall and relaunched the system this year. The Harmony TPV uses pig tissue, … [Read more...] about After recall and relaunch, Medtronic wants to go global with its catheter-delivered Harmony valve
Olympus corrective action covers laser-compatible bronchoscopes
Olympus is warning health providers about incompatible laser use with bronchoscopes after receiving reports of endobronchial combustion, serious patient injury, and one death. The Japanese medtech giant said on July 3 that its voluntary field corrective action involves 32 BF series endoscope models, 19 of which it distributes in the U.S. The … [Read more...] about Olympus corrective action covers laser-compatible bronchoscopes
Teleflex recall of Arrow catheter system is Class I
The FDA today designated Teleflex's recall of its Arrow Endurance extended dwell peripheral catheter system as Class I, the most serious type of medical device recall. The recall involved 262,016 devices distributed from October 26, 2018, to May 10, 2023, according to the FDA. Teleflex and its Arrow International subsidiary have reported 83 … [Read more...] about Teleflex recall of Arrow catheter system is Class I
FDA approves Metavention’s renal denervation for hypertension pivotal IDE study
Metavention announced today that the FDA granted approval to initiate an investigational device exemption (IDE) trial for its renal denervation system. Minneapolis-based Metavention designed its integrated radiofrequency (iRF) renal denervation system to treat hypertension. The company plans for its randomized, double-blinded, sham-controlled … [Read more...] about FDA approves Metavention’s renal denervation for hypertension pivotal IDE study
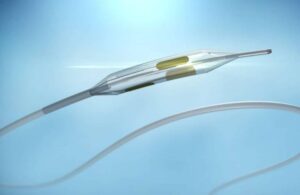
FDA approves Surmodics SurVeil drug-coated balloon
Surmodics (Nasdaq:SRDX) announced today that the FDA granted approval for its SurVeil drug-coated balloon (DCB). Eden Prairie, Minnesota–based Surmodics may now market and sell SurVeil in the U.S. for percutaneous transluminal angioplasty. Use follows appropriate vessel preparation or de novo or restenotic lesions in femoral and popliteal … [Read more...] about FDA approves Surmodics SurVeil drug-coated balloon
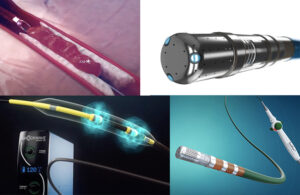
The top catheter innovation news stories of 2023 — so far
The world of catheter innovation has already seen remarkable strides this year— with some of the most promising medtech delivered into the body via catheter-based procedures. Think of improved ablation for treating AFib, coronary intravascular lithotripsy to combat coronary artery disease, and more. Here, we've highlighted the top five … [Read more...] about The top catheter innovation news stories of 2023 — so far
Abbott wins FDA approval for next-gen TactiFlex ablation catheter
Abbott (NYSE: ABT) today announced FDA approval of its TactiFlex Ablation Catheter, Sensor Enabled, which the company describes as the world's first ablation catheter with a flexible tip and contact force technology. The TactiFlex catheter also integrates with Abbott's EnSite X EP System, enabling physicians to visualize heart anatomy more … [Read more...] about Abbott wins FDA approval for next-gen TactiFlex ablation catheter