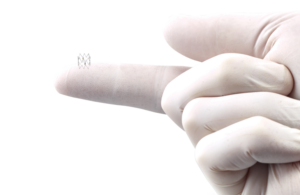
Biosense Webster today launched its HelioStar balloon ablation catheter in Europe. Johnson & Johnson's subsidiary Biosense Webster launched the radiofrequency balloon ablation catheter for catheter-based cardiac electrophysiological mapping of the atria when used with a multi-channel RF generator. According to the company, more than 11 … [Read more...] about Biosense Webster launches HelioStar in Europe
Balloons
Getinge partners with Medtronic to make Radiant balloon-expandable covered stent
Getinge today announced a partnership with Medtronic (NYSE:MDT) over the Radiant balloon-expandable stent, which received CE mark. Radiant is designed for use in chimney endovascular aneurysm repair (ChEVAR) with the Endurant II/IIs stent graft system. It maintains perfusion to renal arteries when used in combination with the stent graft. The … [Read more...] about Getinge partners with Medtronic to make Radiant balloon-expandable covered stent
Advanced NanoTherapies wins FDA breakthrough nod for drug-coated balloon
Advanced NanoTherapies announced today that it received FDA breakthrough device designation for its SirPlux Duo drug-coated balloon (DCB). Los Gatos, California-based Advanced NanoTherapies received the breakthrough nod for the use of the DCB in treating coronary artery disease in vessels less than 3.0mm. The regulatory win comes weeks after two … [Read more...] about Advanced NanoTherapies wins FDA breakthrough nod for drug-coated balloon
LeMaitre Vascular warns on some Syntel Silicone over-the-wire embolectomy catheters
LeMaitre Vascular (NSDQ:LMAT) recently issued an urgent field safety notice for some of its Syntel Silicone over-the-wire embolectomy catheters. The Burlington, Massachusetts-based company issued the warning due to the French size next to the outside diameter symbol on the label not reflecting the 5 Fr catheter extrusion used on all models of … [Read more...] about LeMaitre Vascular warns on some Syntel Silicone over-the-wire embolectomy catheters
BD initiates first-in-human trial of sirolimus drug-coated balloon
BD today announced it started enrollment in the first-in-human trial of a peripheral sirolimus drug-coated balloon (DCB). The Prevision trial is a prospective, multi-center, single-arm, non-randomized study that will evaluate the safety of BD's DCB in the treatment of peripheral arterial disease (PAD) in the femoropopliteal arteries. The company … [Read more...] about BD initiates first-in-human trial of sirolimus drug-coated balloon
The 24 best medical device innovations of 2022
The Galien Foundation recently announced its nominees of medical device innovations for its 2022 Prix Galien USA awards. There are 24 medical technologies nominated for the annual award this year, up from 18 nominees in 2021. The Galien Foundation’s annual Prix Galien awards highlight devices, biotechnology and pharmaceutical products … [Read more...] about The 24 best medical device innovations of 2022
MedAlliance sirolimus-eluting balloon wins second FDA IDE
MedAlliance this week announced its Selution SLR sirolimus-eluting balloon received conditional FDA investigational device exemption (IDE) approval to begin a clinical trial for occlusive disease of the superficial femoral artery (SFA). The second IDE approval comes a few months after the Geneva, Switzerland-based company won IDE approval for … [Read more...] about MedAlliance sirolimus-eluting balloon wins second FDA IDE
Advanced NanoTherapies closes $7.2M Series A for drug-coated balloon
Advanced NanoTherapies announced today that it closed an oversubscribed Series A equity financing round. Los Gatos, California-based Advanced NanoTherapies, which develops the SirPlux Duo drug-coated balloon (DCB) for treating de novo coronary artery disease (CAD) lesions, has now raised $12.5 million in total since September 2020. Get the full … [Read more...] about Advanced NanoTherapies closes $7.2M Series A for drug-coated balloon
Philips announces positive three-year results for its Tack endovascular system
Philips this week announced the latest results from its Tack Optimized Balloon Angioplasty (TOBA) II below-the-knee (BTK) clinical trial. Amsterdam-based Royal Philips designed Tack to provide a sustained treatment effect and positive impact on quality of life for patients with critical limb ischemia (CLI), a severe stage of peripheral arterial … [Read more...] about Philips announces positive three-year results for its Tack endovascular system
Biomerics opens first European research & development facility
Biomerics this week announced it opened a 2,000 sq. ft research and development facility in Galway, Ireland. The new Balloons and Balloons Catheters Center of Excellence will be the first facility in Europe and the second facility outside of the U.S. It will act as an extension to its Athens, Texas operations and will be dedicated to the … [Read more...] about Biomerics opens first European research & development facility