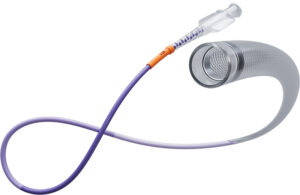
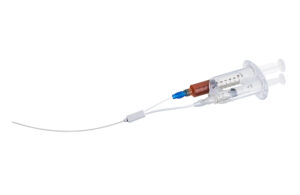
B. Braun Medical announced that it launched its new CareSite Micro luer access device designed to make IV access safer. Bethlehem, Pennsylvania-based B. Braun designed the device to reduce exposure to harmful chemicals. It aims to decrease infection risk in IV access for patients and healthcare providers. The company says its device is the … [Read more...] about B. Braun launches luer device for IV infusions
Catheters
Cerenovus President Mark Dickinson on the future of stroke care
Cerenovus President Mark Dickinson forecasts the innovative technologies that will advance stroke care in the coming years. It's getting harder to beat aspiration systems for fast and simple thrombectomies to remove blood clots that are blocking oxygen from a stroke patient's brain. That's according to Cerenovus Worldwide President Mark … [Read more...] about Cerenovus President Mark Dickinson on the future of stroke care
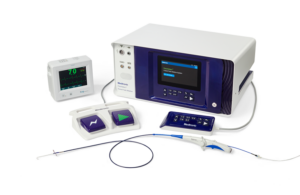
Medtronic PulseSelect pulsed field ablation wins FDA approval
The FDA has approved the Medtronic PulseSelect pulsed field ablation (PFA) system, the device developer said today. PulseSelect is the first PFA technology approved for use in the U.S., as well as the first PFA technology with FDA breakthrough designation to win approval. The minimally invasive, cardiac ablation system is indicated for the … [Read more...] about Medtronic PulseSelect pulsed field ablation wins FDA approval
J&J’s Biosense Webster treats first patients in dual-energy AFib ablation trial
Johnson & Johnson's Biosense Webster today announced the first completed patient cases in a study of its dual-energy ablation catheter. Biosense Webster designed the ThermoCool SmartTouch SF to deliver both radiofrequency (RF) and pulsed-field ablation (PFA) energy. The SmartPulse pivotal study evaluates the dual-energy system in the … [Read more...] about J&J’s Biosense Webster treats first patients in dual-energy AFib ablation trial
Microbot Medical is a step closer to an FDA IDE submission
Microbot Medical today announced the successful completion of its pivotal pre-clinical study of its Liberty endovascular robotic surgical system. The study, performed under good laboratory practice (GLP) and essential for the company's investigational device exemption (IDE) submission, took place under rigorous FDA guidelines. The study involved … [Read more...] about Microbot Medical is a step closer to an FDA IDE submission
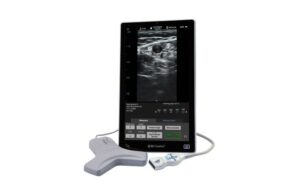
BD launches SiteRite vascular access ultrasound system
BD (NYSE:BD) announced today that it launched its new SiteRite 9 advanced ultrasound system for helping with vascular access device placement. Franklin Lakes, New Jersey-based BD designed the system to help improve clinician efficiency when placing these devices. They include peripherally inserted central catheters (PICCs), central venous … [Read more...] about BD launches SiteRite vascular access ultrasound system
First patient enrolled in Penumbra study of computer-assisted vacuum thrombectomy
Penumbra (NYSE:PEN) today announced the first patient enrolled in STORM-PE, a trial of its Lightning Flash thrombectomy system. Lightning Flash, a mechanical thrombectomy system, received FDA clearance in January. The system features Penumbra’s novel Lightning intelligent aspiration technology with dual-clot detection algorithms. Combining it … [Read more...] about First patient enrolled in Penumbra study of computer-assisted vacuum thrombectomy
VVT Medical announces first commercial ScleroSafe case in U.S.
VVT Medical recently announced the successful completion of the first commercial U.S. varicose vein treatment using its ScleroSafe platform. Dr. Steve Elias performed the procedure at the Center for Vein Disease, part of the Englewood Health Network in Englewood, New Jersey. “The Sclerosafe procedure takes less than 30 minutes, requires only … [Read more...] about VVT Medical announces first commercial ScleroSafe case in U.S.
Medtronic pulsed-field ablation system wins CE mark
Medtronic said today that it's received a CE mark for its PulseSelect pulsed field ablation system. In addition, the medtech giant received European Union approval for its Nitron CryoConsole, which builds upon the legacy of the company's cryo franchise with features to optimize the workflow for cryoballoon ablation. The news of the regulatory … [Read more...] about Medtronic pulsed-field ablation system wins CE mark
Surmodics has positive data on multiple drug-coated balloons
Surmodics (Nasdaq:SRDX) today announced positive study results for two of its drug-coated balloon (DCB) technologies. The company presented data supporting the SurVeil and Sundance DCBs at the 50th Annual VEITH Symposium in New York. In the TRANSCEND trial, Surmodics evaluated the safety and efficacy of SurVeil versus the Medtronic In.Pact … [Read more...] about Surmodics has positive data on multiple drug-coated balloons