This year was a big one for catheter innovation as medtech companies large and small received regulatory approvals and researchers developed catheters that could soon make navigating the vasculature system easier. The numerous innovations weren't just limited to adults. One company launched a cryoablation catheter for children as young as 2 … [Read more...] about The top 10 catheter innovation news stories of 2022
medtronic
Medtronic completes enrollment in pulsed-field ablation catheter trial
Medtronic today announced it completed enrollment and final treatment in the Sphere Per-AF pivotal trial to study Affera's pulsed-field ablation catheter tech. The trial is evaluating the safety and effectiveness of the Sphere-9. The Sphere-9 is a pulsed field (PF) and radio frequency (RF) ablation and high-density mapping catheter. Along with … [Read more...] about Medtronic completes enrollment in pulsed-field ablation catheter trial
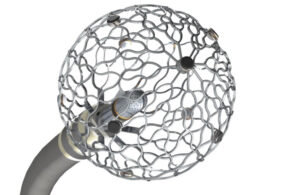
Why Affera’s cardiac ablation technology is worth $1B to Medtronic
Affera started in 2014 with a simple goal that paid off when Medtronic (NYSE:MDT) bought the company for up to $1 billion this year. Achieving that goal, however, took some unconventional and sometimes difficult design choices, Affera founder and CEO Doron Harlev said. Newton, Massachusetts–based Affera's system diagnoses, maps and treats … [Read more...] about Why Affera’s cardiac ablation technology is worth $1B to Medtronic
FDA approves Medtronic drug-eluting stents for treating bifurcation lesions
Medtronic (NYSE:MDT) announced today that the FDA approved its Onyx drug-eluting stents (DES) for treating certain lesions. Approval covers the treatment of non-left main bifurcation lesions utilizing the provisional bifurcation stenting technique. The technique uses a single stent to treat the bifurcation in percutaneous coronary interventions … [Read more...] about FDA approves Medtronic drug-eluting stents for treating bifurcation lesions
FDA labels Medtronic recall of some endotracheal tubes as Class I
The FDA this week said the Medtronic recall of some NIM Contact Reinforced EMG endotracheal tubes is Class I, the most serious kind. Medtronic is recalling its NIM Contact Reinforced EMG endotracheal tubes and NIM Standard Reinforced EMG endotracheal tubes due to reports of obstruction of the endotracheal tube while in use in patients. Tube … [Read more...] about FDA labels Medtronic recall of some endotracheal tubes as Class I
Medtronic warns on Prevail Paclitaxel-coated PTCA balloon catheter
Medtronic this week issues an urgent field safety notice for some of its Prevail Paclitaxel-coated PTCA balloon catheters. The Fridley, Minnesota-based company issued the warning due to the potential for an incorrect compliance chart attached to the catheter hoop. The balloon size on the compliance chart differed from the balloon size detailed … [Read more...] about Medtronic warns on Prevail Paclitaxel-coated PTCA balloon catheter
Medtronic launches Onyx Frontier drug-eluting stent
Medtronic (NYSE:MDT) today launched its newest drug-eluting coronary stent, Onyx Frontier. The Fridley, Minnesota-based company designed the Onyx Frontier drug-eluting stent (DES) to build upon its Resolute Onyx DES. It uses the same stent platform as the Resolute Onyx and has an enhanced delivery system to increase acute performance in … [Read more...] about Medtronic launches Onyx Frontier drug-eluting stent
Medtronic’s Covidien Palindrome and Mahurkar hemodialysis catheter recall is Class I
Medtronic's subsidiary Covidien issued a Class I recall of more than 1 million Palindrome and Mahurkar hemodialysis catheters. Covidien issued the recall due to a catheter hub defect that will connect both extension catheters. The defect could leak within the hub of a chronic dialysis catheter, which can lead to mixing of the arterial and venous … [Read more...] about Medtronic’s Covidien Palindrome and Mahurkar hemodialysis catheter recall is Class I
Design challenges to overcome when developing cardiac ablation devices
It’s about figuring out how and where to go with the cardiac ablation and then engineering the best catheter-based delivery device, according to a top Acutus Medical scientist. Cardiac ablation is when a physician intentionally destroys a small amount of tissue in the heart to treat and prevent heart rhythm problems. The procedure creates … [Read more...] about Design challenges to overcome when developing cardiac ablation devices
What is pulsed-field ablation? Here’s what you need to know
Top experts at Boston Scientific, Medtronic and Acutus Medical shared insights about pulsed-field ablation’s potential at DeviceTalks Boston. Pulsed-field ablation is a non-thermal method for cardiac ablation that has the potential to positively disrupt the way atrial fibrillation is treated. PFA’s roots go back to the dc ablation tech of the … [Read more...] about What is pulsed-field ablation? Here’s what you need to know